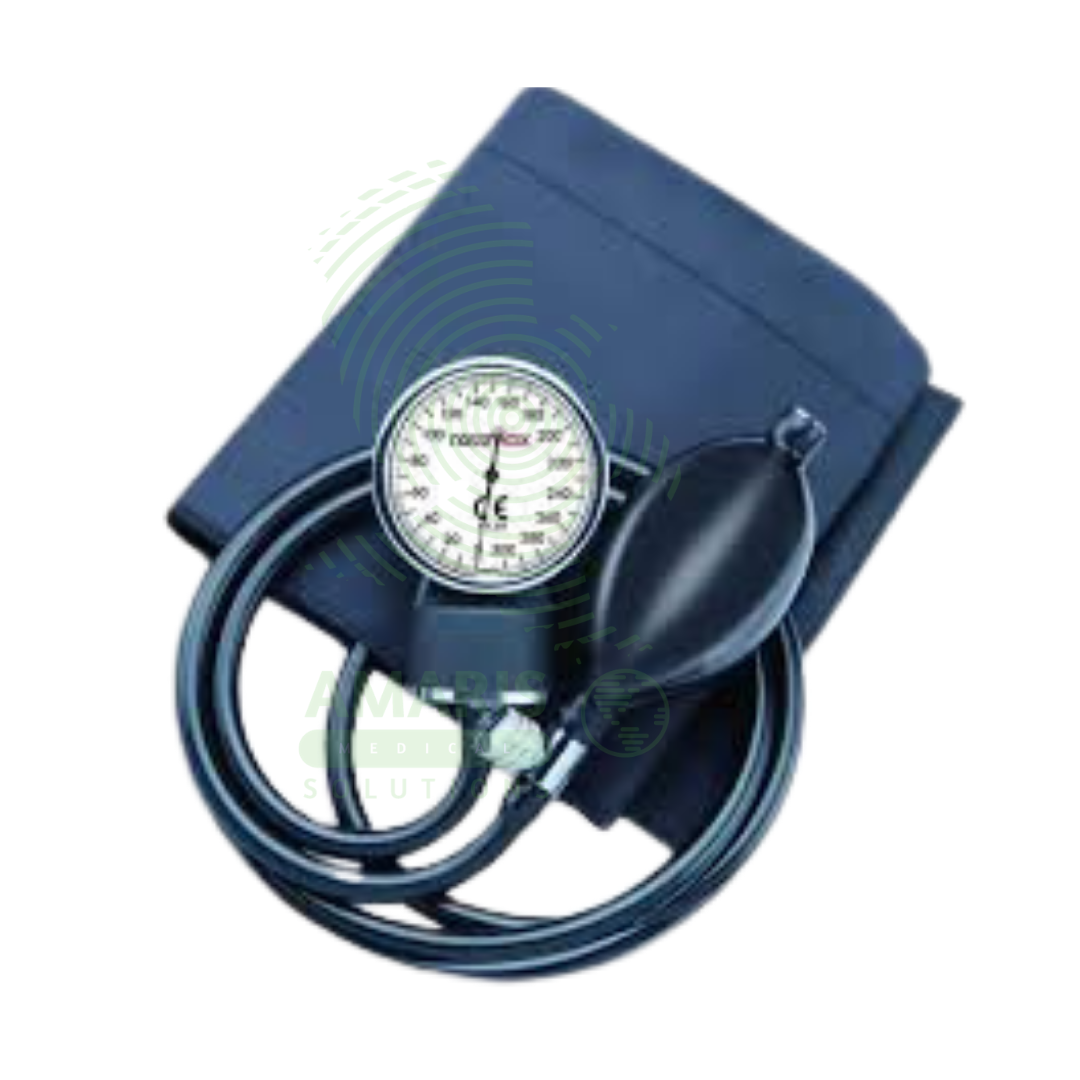
Aneroid Blood Pressure Monitor
WhatsApp Order
The Aneroid Blood Pressure Monitor is a manual, mechanical sphygmomanometer used for the accurate, non-invasive measurement of arterial blood pressure. Operating on the auscultatory method, it consists of an inflatable cuff, a pump bulb with a release valve, and a calibrated dial gauge. Renowned for its reliability, durability, and independence from electrical power, it serves as a clinical gold standard and essential tool in physician offices, hospitals, and for guided home monitoring. Its accuracy is dependent on proper technique, correct cuff size, and regular calibration, making it a fundamental device for diagnosing and managing hypertension and assessing cardiovascular health.
Description
Aneroid Blood Pressure Monitor
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Routine Blood Pressure Screening:
-
Primary Use: The fundamental tool for measuring systolic and diastolic blood pressure during routine physical examinations, health check-ups, and in various clinical settings such as physician offices, outpatient clinics, and community health screenings.
-
How it helps: Provides quick, reliable baseline data that alerts healthcare providers to potential cardiovascular issues early, allowing for timely intervention before minor concerns develop into serious health problems.
2. Diagnosis & Management of Hypertension:
-
Primary Use: Essential for the initial diagnosis, ongoing monitoring, and management of hypertension (high blood pressure). It allows healthcare providers to track treatment efficacy and make necessary medication adjustments.
-
How it helps: Gives doctors the objective data they need to accurately diagnose high blood pressure and fine-tune medications, helping patients achieve healthy levels that significantly reduce their risk of heart attack, stroke, and kidney disease.
3.Pre-operative & Pre-procedural Assessment:
-
Primary Use: A standard component of pre-surgical and pre-procedural evaluations to establish a patient’s baseline blood pressure and assess cardiovascular stability before anesthesia or intervention.
-
How it helps: Ensures patient safety in the operating room by identifying uncontrolled hypertension or other blood pressure abnormalities that could lead to dangerous complications during surgery.
4. Monitoring During Medical Consultations:
-
Primary Use: Used to assess vital signs during patient visits for a wide range of complaints, providing immediate data that can inform clinical decisions related to cardiac, renal, or neurological health.
-
How it helps: Offers doctors a real-time snapshot of cardiovascular health during appointments, helping them connect seemingly unrelated symptoms—like headaches or dizziness—to underlying blood pressure issues.
5. Home Blood Pressure Monitoring (Clinical Guidance):
-
Primary Use: When prescribed or recommended by a clinician, it is used by patients for self-monitoring at home to provide valuable trend data outside the clinical environment, aiding in diagnosis and treatment of “white coat hypertension” or assessing medication control.
-
How it helps: Empowers patients to take an active role in their health by tracking their blood pressure in their natural environment, providing doctors with a more accurate picture than occasional office readings alone.
SECONDARY & SUPPORTIVE USES
1. Public Health & Wellness Campaigns: Widely used in non-clinical settings such as pharmacies, workplace wellness programs, and health fairs for public blood pressure education and preliminary screening, helping identify undiagnosed hypertension in the community.
2. Nursing Home & Long-Term Care Facility Rounds: A durable and reliable tool for daily or weekly vital sign checks on residents in geriatric and long-term care settings, supporting ongoing health monitoring for vulnerable populations.
3. Sports Medicine & Fitness Assessments: Used to monitor blood pressure response during physical activity assessments or as part of overall cardiovascular health evaluations for athletes.
4. Medical & Nursing Education: A fundamental teaching device for instructing students on proper blood pressure measurement technique, Korotkoff sound identification, and cuff placement, building essential clinical skills for future healthcare providers.
5. Backup Device in Clinical Settings: Serves as a critical backup when electronic monitors fail, during power outages, or in situations where electronic interference is a concern, ensuring patient care is never interrupted.
KEY PRODUCT FEATURES
1.BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: Manual Sphygmomanometer (Aneroid Type).
-
Measurement Method: Auscultatory method using a stethoscope and the detection of Korotkoff sounds.
-
Core Components: Consists of an inflatable cuff, a hand-operated bulb pump with a release valve, and a mechanical pressure gauge (aneroid manometer).
-
Output: Direct reading from a calibrated dial gauge (manometer) in millimeters of mercury (mmHg).
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Pressure Range: Typically measures from 0 mmHg to 300 mmHg, covering the full clinical range.
-
Accuracy: High accuracy when properly calibrated, often within ±3 mmHg. Requires periodic calibration against a mercury column or digital standard.
-
Cuff Sizes: Available in multiple sizes (e.g., Adult, Large Adult, Child, Thigh) to ensure accurate measurement. Correct cuff size (bladder width covering 80% of arm circumference) is critical.
-
Gauge Display: Features a large, easy-to-read dial with clear markings and a needle indicator. May include a built-in stethoscope head or require a separate stethoscope.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Form Factor: Portable, handheld, or wall-mounted unit. Often housed in a durable case.
-
Durability: Robust mechanical construction with no electronic components, making it resistant to damage from drops or environmental factors (excluding water immersion).
-
Maintenance: Requires no batteries or power source. Primary maintenance involves checking the integrity of the bladder, tubing, and valve, and ensuring gauge calibration.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Classified as a Class I or Class IIa medical device in most regions (e.g., CE Marked, FDA Cleared). Often exempt from pre-market notification where manual devices are recognized standards.
-
Standards: Manufactured in compliance with standards such as ISO 81060-2 (Non-invasive sphygmomanometers) and general safety standards (e.g., IEC 60601-1 if applicable).
-
Calibration: Must be calibrated at regular intervals (e.g., annually or per manufacturer/regulatory guidelines) to maintain measurement accuracy. A calibration sticker is often present.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage Conditions: Store in a clean, dry, and temperature-stable environment. Avoid extreme heat, cold, or humidity which can affect the rubber components and gauge mechanism.
-
Handling: Protect the gauge from sharp impacts. Release all air from the cuff and loosely coil the tubing after use to prevent kinks and cracks. Store with the control valve open.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Gold-standard, non-invasive measurement of arterial blood pressure in clinical and non-clinical environments.
-
Calibration Reference: Serves as a reference device for validating and calibrating automated electronic blood pressure monitors.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Personal Protective Equipment (PPE): Standard clinical hygiene applies. No special PPE is required for routine use.
-
Patient Safety: Always use the correct cuff size for the patient's arm to prevent inaccurate readings (both falsely high and low). Do not inflate the cuff beyond 300 mmHg or the patient's discomfort level. Apply the cuff over bare skin or very thin clothing.
-
Device Safety: Inspect the device before each use. Check for cracks in the tubing, leaks in the bladder or bulb, and ensure the needle returns to zero when not in use. Do not use if damaged.
2. FIRST AID MEASURES
-
General: The device is non-invasive and low-risk. Any emergency would be related to the patient's underlying condition, not the device.
-
Injury from Device: If over-inflation causes patient discomfort or minor injury (e.g., petechiae), release the cuff immediately. Provide reassurance and simple first aid if needed (e.g., rest, observation).
-
Ingestion of Parts: Not applicable under normal use. Keep small parts (e.g., valve components) away from children.
3. FIRE FIGHTING MEASURES
-
Flammability: The rubber/bladder, plastic casing, and vinyl tubing are combustible. The metal gauge and components are not.
-
Extinguishing Media: Use water, foam, CO₂, or dry chemical extinguishers as appropriate for the surrounding fire.
-
Hazardous Combustion Products: Burning rubber and plastics may release acrid smoke and toxic fumes.
-
Firefighter Instructions: Standard firefighting procedures apply. No unique hazards.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties