Microscope
WhatsApp Order
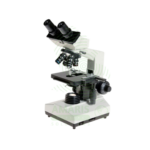
A microscope is a Class I medical device (optical instrument) essential for visualizing microorganisms, cells, and tissues in clinical diagnostics, featuring magnifications from 40× to 1000× (oil immersion) with brightfield, phase contrast, fluorescence, darkfield, or polarized light capabilities. Standard clinical microscopes are binocular or trinocular with 4×, 10×, 40×, and 100× (oil) plan objectives, 10× widefield eyepieces, Abbe condenser, mechanical stage, and halogen or LED illumination. Fluorescence microscopes add specific filter cubes (FITC, TRITC, DAPI) and high-intensity light sources (mercury/xenon) for FISH, immunofluorescence, and AFB detection. Primary clinical applications include microbiological examination (Gram stains, AFB, wet mounts), hematology (differential counts, RBC morphology), histopathology (tissue sections, H&E, special stains), cytology (Pap smears, FNAs), urinalysis (sediment examination), parasitology (malaria, ova, parasites), and fertility (semen analysis). Essential equipment in every clinical laboratory for infectious disease diagnosis, cancer detection, hematological disorder evaluation, and countless other diagnostic applications requiring direct visualization of specimens.
Description
Microscope
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Microbiological Examination and Pathogen Identification:
-
Primary Use: Microscopes are essential for identifying bacteria, fungi, parasites, and other microorganisms in clinical specimens including blood, urine, sputum, cerebrospinal fluid, and tissue samples, enabling diagnosis of infectious diseases and guiding antimicrobial therapy.
-
How it helps: Reveals the invisible organisms causing infections, allowing doctors to choose antibiotics that target the specific bacteria or treat the exact parasite making a patient sick.
2. Hematology and Blood Cell Analysis:
-
Primary Use: Used to perform manual differential white blood cell counts, evaluate red blood cell morphology, identify abnormal cells, and assess platelet morphology in patients with hematological disorders including leukemias, anemias, and thrombocytopenias.
-
How it helps: Gives hematologists a direct view of blood cells, revealing the telltale changes that signal leukemia, the characteristic shapes of sickle cell disease, and the subtle abnormalities that guide diagnosis and treatment.
3. Histopathology and Cytology Tissue Examination:
-
Primary Use: Essential for examining stained tissue sections and cytology specimens to diagnose cancers, inflammatory conditions, and other pathological processes in surgical pathology and cytology laboratories.
-
How it helps: Enables pathologists to see cancer cells in tissue biopsies, identify the extent of disease, and determine whether all abnormal tissue has been removed during surgery.
4. Urinalysis and Sediment Examination:
-
Primary Use: Used to identify cells, casts, crystals, bacteria, and parasites in urine sediment for diagnosis of urinary tract infections, renal diseases, and metabolic disorders.
-
How it helps: Reveals the hidden story in a urine sample, showing doctors whether kidney damage, urinary tract infections, or metabolic disorders are affecting a patient’s health.
5. Fertility and Reproductive Medicine:
-
Primary Use: Employed in semen analysis for sperm count, motility, and morphology assessment in infertility evaluations and assisted reproduction procedures.
-
How it helps: Helps couples understand the factors affecting their fertility, providing essential information that guides treatment decisions and brings them closer to achieving their dream of starting a family.
6. Parasitology and Tropical Medicine:
-
Primary Use: Critical for identifying parasites in blood films (malaria, babesiosis, trypanosomes), stool specimens (ova and parasites), and tissue samples in patients with parasitic infections.
-
How it helps: Spots the parasites that cause devastating tropical diseases, from malaria in blood films to worms in stool samples, ensuring patients receive the right antiparasitic treatment.
7. Cytogenetics and Fluorescence In Situ Hybridization (FISH):
-
Primary Use: Specialized fluorescence microscopes are used to visualize chromosomes and fluorescent probes for genetic abnormality detection in prenatal diagnosis, cancer cytogenetics, and genetic disorders.
-
How it helps: Makes chromosomes glow in specific colors, allowing geneticists to see missing pieces, extra copies, or swapped segments that cause conditions from Down syndrome to certain leukemias.
SECONDARY & SUPPORTIVE USES
1. Research and Clinical Studies: Used in translational research, drug development, and basic science investigations across all medical disciplines, advancing our understanding of disease and treatment.
2. Veterinary Medicine: Essential for diagnostic testing in animal health including blood smears, fecal exams, and tissue analysis, helping veterinarians care for animal patients.
3. Medical Education and Training: Fundamental tool for teaching histology, pathology, microbiology, and hematology to medical students, residents, and laboratory professionals, training the next generation of healthcare providers.
4. Quality Control in Laboratory Testing: Used to verify staining quality, assess specimen adequacy, and validate automated hematology and urinalysis results, ensuring laboratory accuracy.
5. Forensic Medicine: Employed in forensic laboratories for analysis of trace evidence, bloodstains, and tissue samples, helping solve crimes and bring justice to victims.
6. Environmental and Occupational Health: Used to analyze water samples, air samples, and occupational exposure specimens for microorganisms and particulates, protecting public health.
7. Pharmaceutical Quality Control: Essential for particulate analysis, crystal identification, and microbiological testing in pharmaceutical manufacturing, ensuring medications are safe and pure.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: Optical instrument for magnifying and visualizing specimens not visible to the naked eye.
-
Common Names: Microscope, Clinical Microscope, Laboratory Microscope, Medical Microscope, Binocular Microscope, Trinocular Microscope, Fluorescence Microscope, Phase Contrast Microscope.
-
Microscope Types:
-
Brightfield Microscope: Standard for stained specimens (H&E, Gram stain, Pap smear).
-
Phase Contrast Microscope: For observing unstained living cells (cell cultures, wet mounts).
-
Fluorescence Microscope: For fluorescently labeled specimens (FISH, immunofluorescence, AFB).
-
Darkfield Microscope: For visualizing spirochetes and other unstained microorganisms.
-
Polarizing Microscope: For identifying crystals, amyloid, and certain minerals.
-
Inverted Microscope: For examining cell cultures from below (tissue culture, IVF).
-
Dissecting/Stereo Microscope: For low-magnification examination of specimens.
-
-
Optical Configuration:
-
Monocular: Single eyepiece (student/teaching microscopes).
-
Binocular: Two eyepieces (standard clinical microscopes).
-
Trinocular: Two eyepieces plus camera port for photomicrography.
-
-
Magnification Range: 40× to 1000× (standard clinical); up to 1000× with oil immersion.
-
Objectives: 4× (scanning), 10× (low power), 40× (high dry), 100× (oil immersion).
-
Eyepieces: 10× widefield (standard).
-
Illumination: Halogen, LED, or mercury/xenon (fluorescence) light sources.
-
Condenser: Abbe condenser with iris diaphragm for optimal illumination.
-
Stage: Mechanical stage with X-Y controls for precise specimen positioning.
-
Focusing: Coarse and fine focus knobs.
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Optical System: Infinity-corrected or finite-corrected optics; plan (flat-field) objectives for edge-to-edge sharpness.
-
Resolution: Ability to distinguish fine details; limited by numerical aperture of objectives and wavelength of light.
-
Numerical Aperture (NA): Measure of light-gathering ability; higher NA = better resolution (10×: 0.25, 40×: 0.65, 100×: 1.25 oil).
-
Working Distance: Distance between objective and specimen; decreases at higher magnifications.
-
Depth of Field: Thickness of specimen in focus; decreases at higher magnifications.
-
Field of View: Diameter of visible area; decreases at higher magnifications.
-
Parfocality: Objectives remain in focus when rotating nosepiece (minimal adjustment needed).
-
Köhler Illumination: Proper alignment of light path for uniform, glare-free illumination.
-
Fluorescence Capability: Specific filter cubes for different fluorochromes (FITC, TRITC, DAPI, etc.).
-
Camera Compatibility: C-mount or other standard ports for digital cameras.
-
Ergonomics: Adjustable interpupillary distance, diopter adjustment, and tilted eyepieces for comfortable use.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Dimensions: 20-30 cm W × 30-40 cm D × 40-50 cm H (varies by model).
-
Weight: 5-15 kg depending on configuration and materials.
-
Construction: Cast metal base and arm (stability); some modern microscopes use high-strength polymers.
-
Stand: Robust, vibration-dampening design.
-
Focus Mechanism: Coaxial coarse and fine focus knobs with tension adjustment.
-
Nosepiece: Revolving nosepiece with 4-6 objective positions.
-
Stage: Mechanical stage with low-position coaxial controls; stage clips or slide holder.
-
Condenser: Focusable Abbe condenser with centering adjustment.
-
Light Source: Built-in with intensity control; LED long-life or halogen replaceable bulb.
-
Power Requirements: 100-240 VAC, 50/60 Hz or USB-powered for portable models.
-
Certifications: RoHS compliant; CE marked; ISO 9001 manufacturing.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class I medical device (FDA, CE marked for IVD use when used with IVD applications).
-
Electrical Safety: Compliant with IEC 61010-1 for laboratory equipment; low voltage operation.
-
Optical Safety: UV-blocking eyepieces for fluorescence microscopes; safety interlocks for mercury lamps.
-
Chemical Resistance: Stage and frame resistant to common laboratory chemicals and disinfectants.
-
Cleaning: Surfaces designed for easy cleaning with mild detergents and disinfectants.
-
UV Protection: For fluorescence microscopes, UV shields and warning labels.
-
Ergonomics: Designed to reduce operator fatigue during prolonged use.
-
Quality Management: Manufactured under ISO 13485 or ISO 9001 certified processes.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in a clean, dry environment when not in use; use dust cover.
-
Installation: Place on rigid, vibration-free surface; avoid direct sunlight, drafts, and temperature extremes.
-
Cleaning: Clean lenses with lens paper and approved optical cleaner; never use regular tissues or paper. Clean stage and frame with mild detergent and soft cloth.
-
Objective Care: Keep objectives clean; use immersion oil only with oil objectives; clean oil immediately after use.
-
Condenser Care: Keep condenser and filters clean; align per manufacturer instructions.
-
Bulb Replacement: Allow to cool; use specified bulb type; record hours for fluorescence lamps.
-
Annual Maintenance: Professional cleaning, alignment, and calibration recommended.
-
Inspection: Before each use, check objectives, eyepieces, and illumination; clean as needed.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Visual examination of stained and unstained specimens for clinical diagnosis across all laboratory disciplines.
-
Microbiology Applications:
-
Gram Stain: Bacterial morphology and Gram reaction (100× oil).
-
Acid-Fast Stain: Mycobacteria identification (100× oil).
-
Wet Mounts: Motility, fungi, parasites (10×, 40×).
-
Culture Examination: Colony morphology, Gram stain from colonies.
-
-
Hematology Applications:
-
Differential Count: White blood cell classification (50-100 cells, 100× oil).
-
RBC Morphology: Size, shape, color, inclusions (100× oil).
-
Platelet Estimation: Adequacy and morphology (100× oil).
-
Bone Marrow Aspirates: Cell lineage and maturation (100× oil).
-
-
Histopathology Applications:
-
Routine H&E Staining: Tissue architecture and cellular detail (4×, 10×, 40×).
-
Special Stains: Connective tissue, microorganisms, pigments (40×, 100× oil).
-
Immunohistochemistry: Antigen localization with chromogens (10×, 40×).
-
-
Cytology Applications:
-
Pap Smears: Cervical cytology screening (10×, 40×).
-
Fine Needle Aspirates: Cell block and smear examination (40×, 100× oil).
-
Body Fluids: Cell identification (40×, 100× oil).
-
-
Urinalysis Applications:
-
Sediment Examination: Cells, casts, crystals, bacteria (10×, 40×, 100× oil).
-
-
Fluorescence Applications:
-
FISH: Genetic abnormality detection (100× oil, fluorescence filters).
-
Immunofluorescence: Autoantibody detection (40×, fluorescence filters).
-
AFB Fluorescence: Mycobacteria screening (40×, fluorescence filters).
-
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Lens Care: Never touch lenses with fingers; use only lens paper and approved cleaners. Avoid excessive solvent that may damage lens coatings.
-
Oil Immersion: Use only with 100× objectives; clean immediately after use to prevent hardening.
-
Light Source: Halogen and mercury lamps become hot; allow to cool before handling. For fluorescence, record lamp hours and replace per schedule.
-
UV Safety (Fluorescence Microscopes): Never look directly at UV light source; use UV-blocking eyepieces; ensure safety interlocks are functional.
-
Electrical Safety: Keep cords away from water; unplug before cleaning; use only specified voltage.
-
Chemical Safety: Some specimens may contain infectious agents; follow universal precautions; clean spills immediately.
-
Ergonomics: Maintain good posture; adjust eyepieces and stage height for comfort; take regular breaks.
-
Vibration: Place on vibration-free surface; avoid traffic areas; use anti-vibration tables if needed.
-
Alignment: Proper Köhler illumination essential for optimal image quality; realign after bulb changes.
-
Training: Operators should be trained on proper microscope use, care, and cleaning procedures.
2. FIRST AID MEASURES
-
Eye Contact with UV Light (Fluorescence): If accidentally exposed, rest eyes; seek medical attention if symptoms persist.
-
Chemical Splash (Lens Cleaner): Flush eyes with copious water for 15 minutes; seek medical attention.
-
Broken Slide or Cover Glass: Carefully remove fragments with forceps; dispose in sharps container; clean stage and objectives carefully.
-
Specimen Spill on Microscope: Disconnect power; carefully clean with appropriate disinfectant; dry thoroughly before reuse.
-
Mercury Lamp Breakage (Fluorescence): Evacuate area; ventilate; follow mercury spill protocol; use specialized cleanup.
3. FIRE FIGHTING MEASURES
-
Flammability: Plastic components and immersion oil are combustible; metal parts non-combustible.
-
Extinguishing Media: For electrical fire, use CO₂ or dry chemical (Class C) extinguisher.
-
Power Off: Disconnect power if safe to do so.
-
Mercury Lamp: If involved in fire, may release toxic mercury vapor; use SCBA in enclosed spaces.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties