Vacutainers
WhatsApp Order
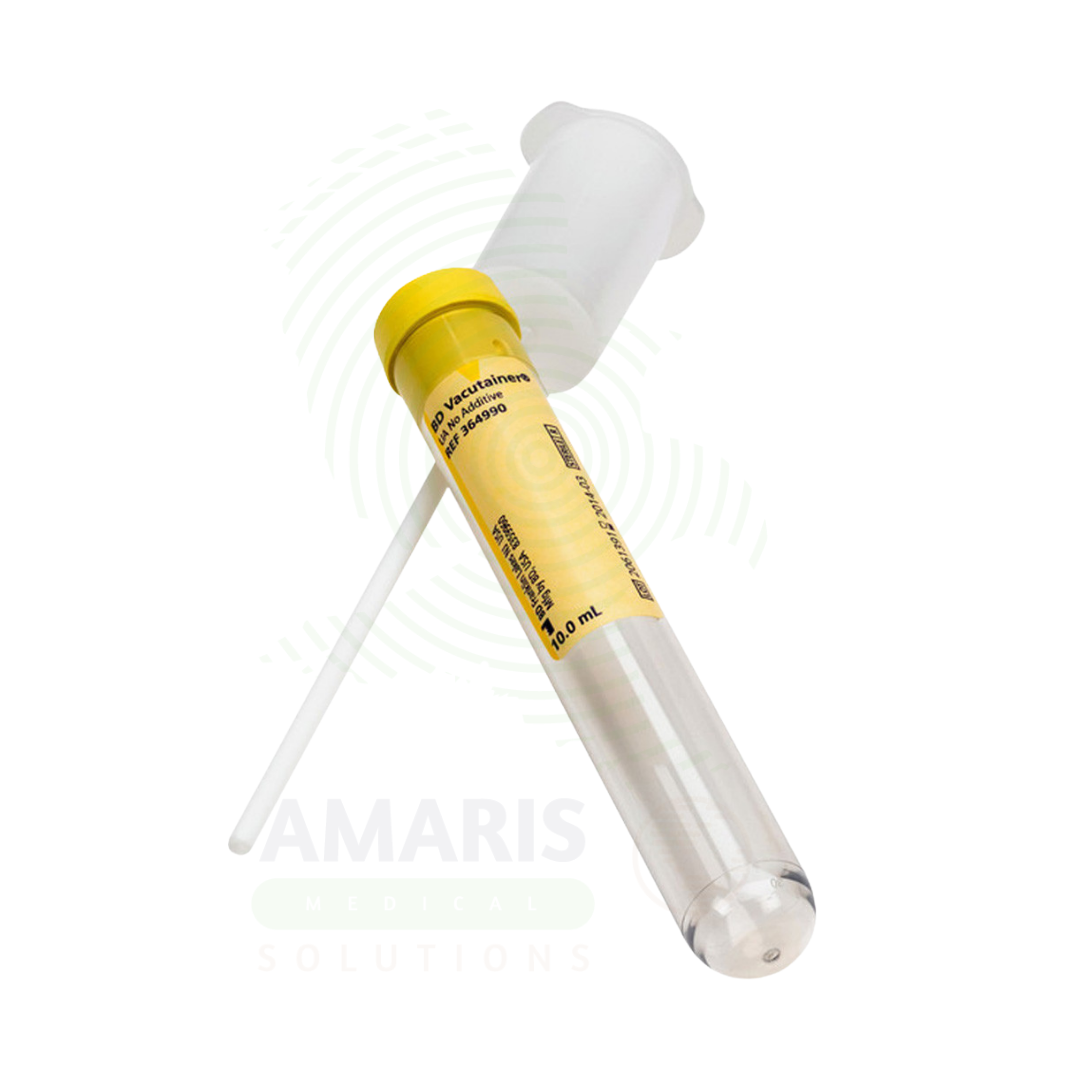
Vacutainers are sterile, single-use, evacuated blood collection tubes used for the collection, transport, and processing of venous blood specimens for laboratory diagnostics. Each tube contains a pre-measured vacuum that draws a precise volume of blood and, in most tubes, a specific additive (anticoagulant, clot activator, preservative, separation gel, or culture media) that preserves specimen integrity for the required analysis. International color-coding of tube closures (e.g., lavender-EDTA, light blue-citrate, gold-serum separator) enables immediate identification of additive and intended use. Available in glass or shatter-resistant PET plastic, with draw volumes ranging from 2 mL to 10 mL. Strict adherence to the CLSI-recommended order of draw, complete fill volume, gentle mixing, and immediate centrifugation are essential for specimen quality and accurate test results. An indispensable device in modern laboratory medicine, clinical practice, and blood banking.
Description
Vacutainers
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Blood Collection for Laboratory Diagnostics
-
Primary Use: Collects, contains, transports, and processes venous blood specimens for a wide range of laboratory analyses, including complete blood count, chemistry panels, coagulation studies, serology, immunohematology, and microbiology. The evacuated tube system uses pre-measured vacuum pressure to draw a precise volume of blood directly from the vein into the tube through a double-ended needle, ensuring consistent blood-to-additive ratios and specimen quality.
-
How it helps: For the phlebotomist, laboratory technician, and ordering physician, the Vacutainer system transforms blood collection from a variable, error-prone process into a standardized, reliable procedure—ensuring that each tube contains exactly the right volume of blood, properly mixed with the correct additive, and ready for accurate analysis. For the patient, this standardization means fewer repeat blood draws, more accurate test results, and confidence that their diagnostic information is reliable.
2. Additive-Specific Blood Preservation
-
Primary Use: Different Vacutainer tubes contain specific additives that prepare blood specimens for specific diagnostic tests, with the vacuum ensuring immediate mixing of blood with the additive upon collection, critical for test accuracy and specimen integrity.
-
How it helps: For the laboratory scientist performing specialized testing, the color-coded additive system ensures that each blood sample arrives in the correct formulation for its intended use—tubes with anticoagulants for plasma tests, tubes with clot activators for serum tests, tubes with preservatives for specialized assays, and tubes with culture media for infection detection. For the patient, this means one blood draw can yield specimens for multiple tests, each collected in the tube that will preserve its unique characteristics for accurate analysis.
3. Standardization of Blood Collection
-
Primary Use: The evacuated tube system standardizes blood collection across healthcare settings, ensuring consistent specimen volume, correct blood-to-additive ratio, and uniform processing conditions, reducing pre-analytical errors, improving laboratory efficiency, and enhancing patient safety.
-
How it helps: For the healthcare system and laboratory quality manager, the standardized Vacutainer system eliminates countless variables that once plagued blood testing—incorrect blood volumes, wrong additives, improper mixing, and inconsistent handling. For the patient, this standardization means their test results are comparable across different facilities and over time, supporting accurate diagnosis and consistent monitoring of chronic conditions.
4. Pediatric and Geriatric Blood Collection
-
Primary Use: Low-volume Vacutainer tubes are available for pediatric, geriatric, and difficult-vein patients, reducing the risk of iatrogenic anemia from excessive blood draw while providing sufficient specimen volume for essential testing.
-
How it helps: For the phlebotomist drawing blood from a premature infant, a frail elderly patient, or an adult with difficult veins, low-volume tubes mean they can obtain the necessary specimens without removing more blood than the patient can safely spare—reducing the risk of anemia from repeated blood draws. For the smallest and most vulnerable patients, these tubes ensure that necessary testing does not become a source of harm.
5. Blood Culture Collection
-
Primary Use: Specialized Vacutainer tubes with culture media formulations are used for the collection of blood for microbiological culture and sensitivity testing, enabling detection of bacteremia and fungemia with reduced contamination risk.
-
How it helps: For the microbiologist and infectious disease physician, blood culture bottles are the gold standard for detecting life-threatening bloodstream infections—the specialized media supports the growth of bacteria and fungi while suppressing contaminants, allowing for accurate identification of pathogens and determination of antibiotic susceptibility. For the patient with sepsis or suspected bloodstream infection, a properly collected blood culture can identify the causative organism and guide life-saving antibiotic therapy.
SECONDARY & SUPPORTIVE USES
1. Quality Control and Proficiency Testing: Used in clinical laboratories for the collection and processing of quality control specimens and proficiency testing samples to ensure accuracy and reliability of diagnostic testing. For the laboratory director, these specimens ensure that testing processes are performing correctly.
2. Research and Clinical Trials: Employed in clinical research settings for the collection and preservation of blood specimens for biomarker analysis, pharmacokinetic studies, and translational research applications. For the researcher, standardized collection ensures data integrity.
3. Blood Donor Screening: Utilized in blood donation centers for the collection of donor blood specimens for mandatory infectious disease testing and blood typing. For the blood recipient, this screening ensures the safety of donated blood.
4. Point-of-Care Interface: Some Vacutainer systems interface with point-of-care testing devices, allowing for direct sampling from the evacuated tube for rapid diagnostic testing. For the clinician needing immediate results, this interface speeds the testing process.
5. Veterinary Medicine: Widely used in veterinary practice for blood collection from companion animals and livestock, with species-specific reference ranges and additive requirements. For the family pet, the same reliable collection system used in human medicine ensures accurate diagnostic testing.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: A sterile, single-use, evacuated glass or plastic tube with a pre-measured vacuum and, in most cases, a specific additive, used for the collection of venous blood specimens.
-
Designation: Defined by additive type, tube size (volume) , closure color, material composition, and sterility status.
-
Core Components:
-
Tube Body: Cylindrical glass or medical-grade plastic (polyethylene terephthalate - PET) container with pre-measured vacuum.
-
Closure: Stopper/septum manufactured from elastomeric material (butyl rubber, bromobutyl rubber, synthetic isoprene) that reseals after needle withdrawal.
-
Additive: Pre-measured liquid or dry chemistry within the tube (anticoagulant, clot activator, preservative, separation gel, culture media).
-
Label Area: Permanent, writable surface for patient identification, date, time, and phlebotomist initials.
-
Safety Closure: Some tubes feature safety-engineered closures or shields to reduce blood exposure risk during transport and processing.
-
-
Core Additive Variants (Color-Coded per CLSI H1-A8 and ISO 6710 Standards):
-
Lavender/Lilac: EDTA (anticoagulant) - Hematology, blood banking.
-
Light Blue: Sodium citrate (anticoagulant) - Coagulation studies (PT, PTT, D-dimer).
-
Red (Plain): No additive or clot activator - Serology, blood banking, chemistry.
-
Gold/Tiger Top: Serum separator gel + clot activator - Routine chemistry.
-
Green: Sodium heparin, lithium heparin (anticoagulant) - Stat chemistry, ammonia, lactate.
-
Gray: Sodium fluoride + potassium oxalate (glycolytic inhibitor + anticoagulant) - Glucose, lactate.
-
Yellow: Acid citrate dextrose (ACD) - HLA typing, DNA studies; or culture media - Microbiology.
-
Royal Blue: Trace element-free (lead, cadmium, mercury) - Toxicology, heavy metal analysis.
-
Tan: Lead-free - Lead testing.
-
Black: Citrate (pediatric coagulation) - Neonatal coagulation studies.
-
Pink: EDTA (blood banking) - Immunohematology, donor screening.
-
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Vacuum Accuracy: The pre-measured vacuum must draw the exact specified fill volume (e.g., 2.0 mL, 3.5 mL, 4.5 mL, 6.0 mL, 8.5 mL, 10.0 mL) to maintain the correct blood-to-additive ratio. Underfill or overfill compromises test results.
-
Additive Precision: Additives are pre-measured to within ±5-10% of target concentration and must be uniformly distributed throughout the tube wall or bottom.
-
Closure Integrity: The elastomeric closure must:
-
Maintain tube vacuum for up to 24-36 months.
-
Reseal completely after needle withdrawal to prevent leakage.
-
Withstand centrifugation forces without leakage or aerosol generation.
-
Be pierceable with minimal coring (fragmentation).
-
-
Centrifugation Compatibility: Tubes must withstand centrifugal forces of 1000-3000 RCF without deformation, leakage, or additive disruption.
-
Separation Gel Performance: For gel-separator tubes, the thixotropic polymer gel must migrate to form a stable barrier between serum/plasma and cells during centrifugation, maintaining barrier integrity during transport and storage.
-
Clot Activation Time: Clot activator-coated tubes must achieve complete clot retraction within 30 minutes to permit timely centrifugation and processing.
-
Shelf Life: Maintain vacuum and additive integrity for 12-24 months (glass) or 18-36 months (PET plastic) from date of manufacture.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Tube Material:
-
Glass (Borosilicate): Traditional material, impermeable to gases, longest vacuum retention, maintains additive stability. Breakage risk.
-
PET Plastic (Polyethylene Terephthalate): Lightweight, shatter-resistant, clear, comparable performance to glass, longer shelf life, reduced sharps injury risk from breakage. Industry standard.
-
-
Tube Dimensions: Vary by volume; typical 13 mm x 75 mm (2-4 mL) to 16 mm x 100 mm (6-10 mL).
-
Closure Color: International, standardized color coding per CLSI and ISO standards for immediate additive identification.
-
Closure Material: Bromobutyl rubber, synthetic polyisoprene, or thermoplastic elastomers. Latex-free.
-
Draw Volume: Actual blood volume collected (typically 10-20% less than total tube capacity due to additive volume).
-
Label Area: Frosted or writable surface; barcode-friendly for laboratory information system integration.
-
Packaging: Sterile, individually sealed in blister packs or plastic pouches; bulk-packed in dispensing trays or vacuum-sealed bags.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Standards:
-
ISO 6710: Single-use containers for venous blood specimen collection.
-
CLSI H1-A8: Procedures and devices for the collection of diagnostic blood specimens by venipuncture.
-
CLSI H3-A6: Procedures for the collection of diagnostic blood specimens by venipuncture.
-
ISO 11137/11135: Sterilization validation (gamma irradiation, ethylene oxide).
-
FDA 510(k) Clearance: Required for US marketing as Class II medical device.
-
CE Marking: Required for European market.
-
IATA/UN 3373: Certification for transport of diagnostic specimens.
-
-
Sterility: Terminally sterilized via gamma irradiation (PET) or ethylene oxide (select glass tubes). Sterility assurance level (SAL) of 10⁻⁶.
-
Biocompatibility: All blood-contact materials must meet ISO 10993 standards for cytotoxicity, sensitization, irritation, and systemic toxicity. Non-pyrogenic, endotoxin-free.
-
Latex-Free: All closures and components manufactured without natural rubber latex.
-
DEHP-Free: PET plastic tubes are DEHP-free; some glass tubes may use DEHP-containing stoppers; non-DEHP alternatives increasingly mandated for neonatal/pediatric applications.
-
Evacuation Integrity: Must maintain vacuum for labeled shelf life; verified via random-sample testing per ISO 6710.
-
Additive Safety: All additives must be USP/EP grade, non-cytotoxic, and certified free of interfering substances.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in original, unopened packaging in a cool, dry environment (15-25°C / 59-77°F). Protect from direct sunlight, extreme temperatures, and humidity. Do not freeze.
-
Shelf Life: Printed on each outer carton and individual tube. Typical: 18-36 months from manufacture date. Do not use after expiration; vacuum loss, additive degradation, and sterility cannot be guaranteed.
-
Package Inspection: Before use, inspect individual packaging for any signs of compromise: tears, punctures, moisture ingress, or damage to sterile barrier. Inspect tube for cracks, visible particulate, or additive discoloration. Do not use it if integrity is questionable.
-
Pre-Analytical Stability: After collection, maintain tubes at appropriate temperature (room temperature, refrigerated, or frozen per test requirements) and protect from light for light-sensitive analytes (bilirubin, vitamin A, vitamin B6, folate, carotene).
-
Centrifugation Protocol: Follow manufacturer specifications for time, speed, and temperature. Inadequate centrifugation causes incomplete separation; excessive centrifugation causes hemolysis and cellular fragmentation.
-
Single-Use Protocol: Vacutainers are strictly single-use devices. Never reuse a Vacutainer tube. Never transfer blood from one tube to another. Never pool specimens. Reuse is associated with:
-
Incorrect blood-to-additive ratio.
-
Clot formation and hemolysis.
-
Cross-contamination and false-positive results.
-
Loss of sterility and infection risk.
-
Compromised vacuum and incomplete fill.
-
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: The universal, indispensable device for venous blood collection across all healthcare settings: hospitals, clinics, physician offices, reference laboratories, blood donation centers, and clinical research facilities.
-
Selection Criteria (Order of Draw): Per CLSI H3-A6, tubes must be collected in a specific sequence to prevent cross-contamination of additives:
-
Blood Culture Bottles (Sterile, no additive)
-
Light Blue (Sodium citrate; coagulation)
-
Red/Gold/Tiger Top (Clot activator/serum)
-
Green (Heparin; plasma chemistry)
-
Lavender/Pink (EDTA; hematology/blood banking)
-
Gray (Fluoride; glucose)
-
Yellow (ACD; HLA/DNA studies)
-
-
Specimen Rejection Criteria: Common causes for laboratory rejection:
-
Unlabeled or mislabeled tube.
-
Expired tube.
-
Underfilled or overfilled tube (affects blood-to-additive ratio).
-
Hemolyzed, lipemic, or icteric specimen.
-
Clotted specimen (anticoagulant tubes).
-
Improper transport temperature.
-
Prolonged transport time.
-
Leaking tube or compromised closure.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Standard Precautions (Most Important): Blood is potentially infectious. Treat all specimens as capable of transmitting bloodborne pathogens (HIV, HBV, HCV). Wear appropriate personal protective equipment (gloves, gown, face shield) during collection and handling.
-
Order of Draw Compliance: Strict adherence to the CLSI-recommended order of draw prevents additive carryover and erroneous test results. Potassium EDTA contamination of sodium citrate tubes causes false coagulation results; clot activator carryover into heparin tubes causes false electrolyte results.
-
Fill Volume Compliance: Tubes must fill to completion. Underfilled tubes alter blood-to-additive ratio: citrate tubes (false prolonged PT/PTT), EDTA tubes (cell shrinkage, false hematocrit), fluoride tubes (inadequate glycolysis inhibition). Do not decant from one tube to another.
-
Do Not Pre-Mix Additives: Some tubes (e.g., citrate, EDTA) require gentle inversion 8-10 times immediately after collection. Do not shake vigorously; it causes hemolysis and foam. Do not delay mixing; it causes microclot formation.
-
Closure Safety: Never remove the closure from a Vacutainer tube containing blood. Aerosolization of blood droplets occurs during decapping, creating biohazard exposure risk. Use safety-engineered transfer devices for aliquotting.
-
Centrifuge Safety: Always balance centrifuge loads. Use sealed carriers or safety cups when centrifuging blood tubes to contain aerosols from tube failure. Do not open the centrifuge lid until the rotor has come to complete stop.
-
Sharps Safety: Used blood collection needles are sharps. Discard immediately into a puncture-resistant sharps container. Never recap, bend, break, or manually remove needles. Activate safety mechanisms immediately after use.
-
Pediatric/Neonatal Considerations: Use low-volume draw tubes to prevent iatrogenic anemia. Verify volume requirements with the laboratory before collection. Some additives (e.g., heparin) require adjustment for neonatal sample volumes.
-
Transport Safety: Package tubes in leak-proof secondary containers with absorbent material for transport. Follow IATA/UN 3373 regulations for shipping diagnostic specimens.
2. FIRST AID MEASURES
-
Needlestick Injury: Immediately wash the affected area vigorously with soap and water. Do not squeeze or suck wounds. Report incident immediately to supervisor/occupational health. Follow institutional post-exposure prophylaxis protocols for bloodborne pathogen exposure (HIV, HBV, HCV). Complete incident documentation.
-
Blood Spill: Contain spill with absorbent material. Clean area with EPA-registered hospital disinfectant effective against bloodborne pathogens. Discard contaminated materials in biohazard waste. Change gloves and wash hands thoroughly after cleanup.
-
Tube Breakage During Centrifugation: Stop centrifuge immediately. Do not open the lid for 30 minutes to allow aerosolized particles to settle. Wear heavy-duty gloves, gown, and face shield during cleanup. Carefully remove tube fragments and contents. Disinfect centrifuge interior and rotors per manufacturer instructions.
-
Chemical Exposure (Additives): Accidental ingestion of tube additives is unlikely due to low volume. If skin or eye contact occurs, rinse with copious water for 15 minutes. Seek medical attention if irritation persists.
-
Allergic Reaction to Closure Material: Rare. If localized contact dermatitis occurs in the phlebotomist's fingers, discontinue use of that product and substitute with alternative closure material. Seek occupational health evaluation.
3. FIRE FIGHTING MEASURES
-
Flammability: PET plastic tubes are combustible; glass tubes are non-combustible. Elastomeric closures are combustible.
-
Extinguishing Media: Use water, foam, CO₂, or dry chemical powder as appropriate for the surrounding fire. Burning plastic releases toxic fumes; use self-contained breathing apparatus (SCBA) in enclosed spaces.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties