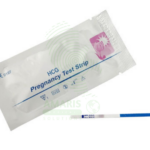
H. pylori Test Strips
WhatsApp Order
H. pylori Test Strips are rapid, lateral flow immunochromatographic devices used for the qualitative detection of Helicobacter pylori antigens in human stool. As a non-invasive stool antigen test (SAT), it provides a convenient and reliable method for diagnosing active H. pylori infection and confirming eradication after therapy, with results available in under 15 minutes. Ideal for use in clinics and outpatient settings, it aids in the management of dyspepsia and peptic ulcer disease. Proper sample handling, adherence to pre-test medication restrictions, and strict biohazard precautions are essential for obtaining accurate results.
Description
H. pylori Test Strips
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Point-of-Care Screening for Helicobacter pylori Infection:
-
Primary Use: The primary use is for the rapid, qualitative detection of specific antigens from Helicobacter pylori in human stool samples. It serves as a convenient, non-invasive screening tool to identify current active infection in patients with symptoms of dyspepsia, gastritis, or peptic ulcer disease.
-
How it helps: Provides doctors with a simple, non-invasive way to check for a common cause of stomach pain, bloating, and ulcers, allowing treatment to begin quickly without subjecting patients to uncomfortable procedures.
2. Non-Invasive Diagnosis in Pediatric and Adult Patients:
-
Primary Use: Particularly useful for patients who cannot or prefer not to undergo invasive procedures like endoscopy and biopsy (the gold standard). It is well-suited for children and for initial workup in primary care settings.
-
How it helps: Spares children and anxious adults from the stress and discomfort of endoscopic procedures, offering a gentle, stool-based test that is much easier for patients of all ages to tolerate.
3. Confirmation of Eradication Post-Treatment:
-
Primary Use: A key application is to confirm the success of H. pylori eradication therapy. A follow-up test is typically performed at least 4 weeks after completion of antibiotic and proton pump inhibitor (PPI) treatment to verify that the infection has been cleared.
-
How it helps: Provides patients and their doctors with clear evidence that the course of antibiotics worked, offering peace of mind that the bacteria has been eliminated and reducing the risk of ulcer recurrence or complications.
4. Epidemiological and Public Health Studies:
-
Primary Use: Used in community health settings and research to estimate the prevalence of H. pylori infection in populations.
-
How it helps: Helps public health officials understand how common this infection is in different communities, guiding efforts to reduce its spread and associated health problems like stomach cancer.
SECONDARY & SUPPORTIVE USES
1. Monitoring High-Risk Groups: Screens individuals with a family history of gastric cancer or those on long-term NSAID therapy who may be at increased risk for H. pylori-related complications, catching infections early when they are easiest to treat.
2. Triaging Patients for Endoscopy: A positive stool antigen test in a symptomatic patient provides a strong indication for upper gastrointestinal endoscopy to assess for ulcers, gastritis, or other pathology, helping doctors prioritize which patients need invasive procedures.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: Rapid, Immunochromatographic, Lateral Flow Assay.
-
Common Name: H. pylori Stool Antigen Test (SAT).
-
Sample Type: Fresh human stool sample. Some kits may also be validated for use with stool samples stored in specific transport media.
-
Target: Detects H. pylori antigens (proteins), specifically indicating an active, current infection (as opposed to antibody tests which can remain positive after past infection).
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Test Principle: Utilizes monoclonal or polyclonal antibodies specific to H. pylori antigens. The stool sample is mixed with an extraction buffer. When this mixture is applied to the test device, it migrates laterally. If H. pylori antigens are present, they bind to antibody-colored conjugate particles, forming a visible colored line in the test region (T).
-
Procedure: A small stool sample is collected and mixed with the provided extraction buffer. Several drops of the mixture are then added to the sample well of the test cassette. Results are read after 5-15 minutes.
-
Interpretation:
-
Positive: Two colored lines appear—one in the Control line region (C) and one in the Test line region (T).
-
Negative: Only one colored line appears in the Control line region (C). No line appears in the Test line region.
-
Invalid: No line appears in the Control region. The test must be repeated with a new device.
-
-
Accuracy: Performance must meet regulatory standards. Sensitivity and specificity are typically >90% when compared to reference methods (histology, urea breath test). Exact figures are kit-dependent and should be verified from the product insert.
-
Time to Result: Results are available in 5 to 15 minutes.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Kit Format: Sold as a complete test kit, usually containing:
-
Test cassettes or strips (individually sealed in foil pouches with desiccant).
-
Sample collection devices (spatula, tube).
-
Extraction buffer vials.
-
Disposable droppers.
-
Instructions for Use (IFU).
-
-
Storage: Most kits require storage at room temperature (2-30°C). Must be kept in a dry place and protected from direct sunlight and freezing. Always check the expiry date before use.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Approvals: Must carry relevant medical device approvals: CE Marking (for EU), FDA 510(k) Clearance (for US), and other national regulatory approvals.
-
Quality Standards: Manufactured under a ISO 13485 quality management system.
-
Intended Use: Approved for professional use in clinics, laboratories, or for prescription home use with proper instructions.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store unopened test kits at the temperature specified by the manufacturer (usually room temperature). Do not use expired kits.
-
Stool Sample Handling: Stool samples should be tested as soon as possible after collection (ideally within 24 hours if refrigerated at 2-8°C). Some kits provide specific transport media for longer stability.
-
Waste Disposal: The used test device, sample collection materials, and gloves should be disposed of as biohazardous waste following local regulations.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: A rapid, non-invasive screening and diagnostic tool for active H. pylori infection, suitable for use in outpatient clinics, physician offices, and smaller laboratories.
-
Important Consideration: Proton Pump Inhibitors (PPIs), antibiotics, and bismuth compounds can suppress bacterial load and lead to false-negative results. Patients should ideally stop PPIs for 2 weeks and antibiotics/bismuth for 4 weeks prior to testing.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Biohazard Precautions: Stool samples are potential sources of infection. Perform testing wearing appropriate Personal Protective Equipment (PPE): disposable gloves, lab coat, and safety glasses if splashing is possible.
-
Avoid Contamination: Do not touch the sample window (membrane) of the test cassette. Use the provided dropper for sample application.
-
Follow Instructions Precisely: Adhere strictly to the incubation times and sample preparation steps in the IFU. Adding too much or too little sample can affect the result.
-
Interpretation Window: Read results within the specified time frame (e.g., at 10 minutes). Do not interpret results after 20-30 minutes, as an evaporation line may appear and be misinterpreted as positive.
2. FIRST AID MEASURES
-
Ingestion: If any kit components (buffer, test device) are ingested, seek medical advice and provide the product information.
-
Skin/Eye Contact: If extraction buffer contacts skin or eyes, rinse immediately with plenty of water for several minutes. If irritation persists, seek medical advice.
3. FIRE FIGHTING MEASURES
-
Flammability: The plastic cassette, paper components, and packaging are combustible.
-
Extinguishing Media: Use water, foam, or CO₂ as appropriate for the surrounding fire.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties