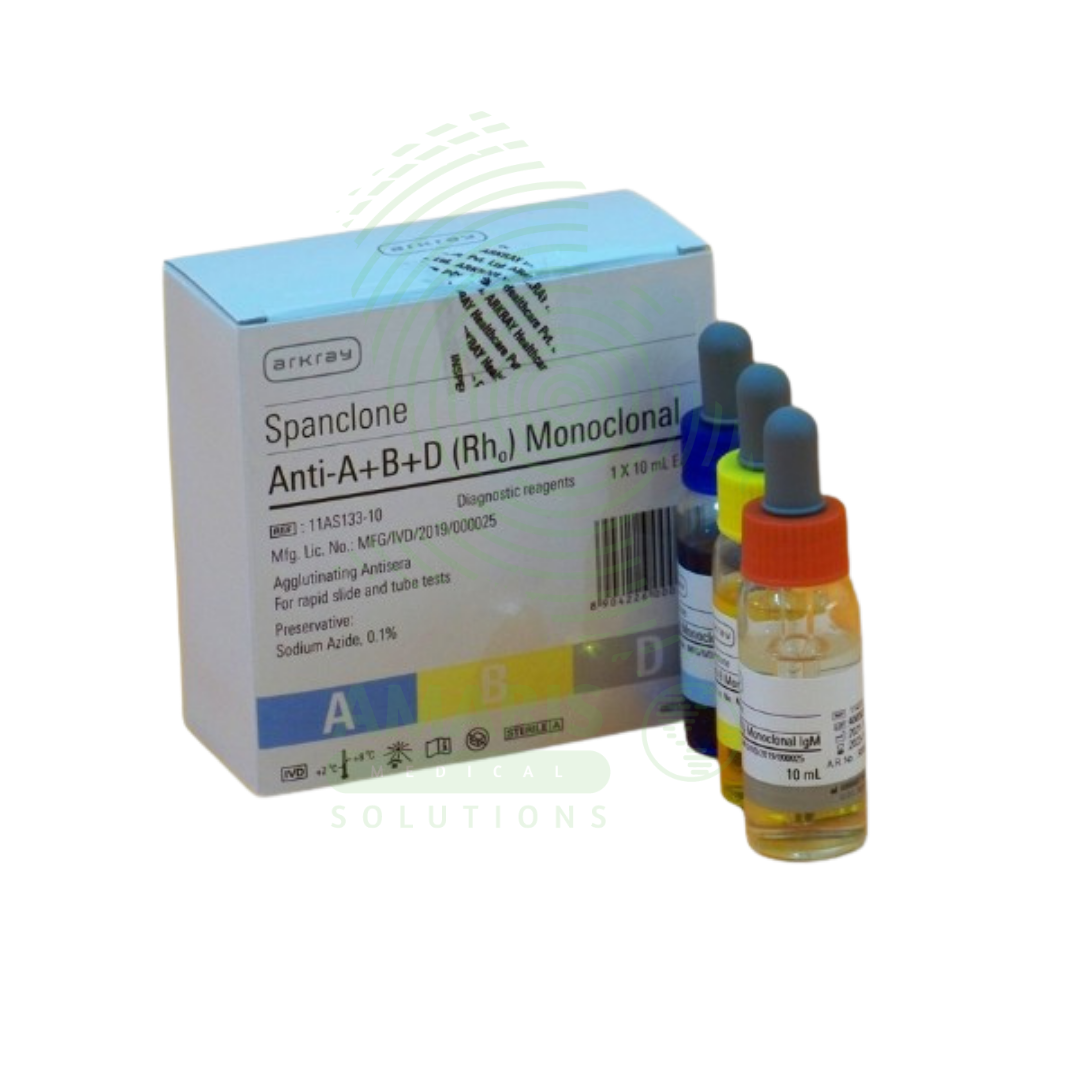
Blood Grouping kit
WhatsApp Order
A Blood Grouping Kit is a Class II medical device (FDA-cleared/CE-marked) containing monoclonal antibodies (Anti-A, Anti-B, Anti-D) and control reagents for determining ABO blood group and Rh factor by agglutination method. The kit includes color-coded reagent vials, reaction cards or slides, mixing sticks, lancets, and capillary pipettes. Test time 2-5 minutes (slide method) or 15-30 minutes (tube method). Interpretation based on agglutination pattern: Anti-A positive = group A; Anti-B positive = group B; both positive = group AB; both negative = group O; Anti-D positive = Rh positive; Anti-D negative = Rh negative. Primary clinical applications include pre-transfusion compatibility testing, prenatal and neonatal testing (Rh incompatibility prevention), preoperative blood typing, blood donor screening, and emergency blood typing. Critical safety precautions include refrigerated storage (2-8°C), proper disposal of lancets in sharps containers, universal precautions for blood samples, and confirmation with reverse typing when possible. Essential kit for transfusion medicine, obstetrics, surgery, and emergency care.
Description
Blood Grouping kit
PRIMARY CLINICAL & DIAGNOSTIC USES
1. ABO and Rh Blood Typing:
-
Primary Use: Used to determine an individual’s ABO blood group (A, B, AB, or O) and Rh factor (positive or negative), which is essential for blood transfusion compatibility, organ transplantation, and prenatal care.
-
How it helps: Provides the foundational information needed for safe blood transfusions, ensuring patients receive blood that matches their own type and preventing life-threatening immune reactions that can occur with mismatched blood.
2. Pre-Transfusion Compatibility Testing:
-
Primary Use: Ensures donor and recipient blood types are compatible before blood transfusion, preventing potentially fatal hemolytic transfusion reactions caused by ABO incompatibility.
-
How it helps: Acts as a final safety check before blood is administered, giving doctors and patients confidence that the transfusion will be safe and effective rather than causing a dangerous immune response.
3. Prenatal and Neonatal Testing:
-
Primary Use: Used to determine blood type of pregnant women and assess risk of Rh incompatibility with fetus, guiding administration of Rh immunoglobulin (RhoGAM) to prevent hemolytic disease of the newborn.
-
How it helps: Protects future pregnancies by identifying Rh incompatibility early, allowing doctors to administer preventive treatment that stops the mother’s immune system from attacking red blood cells of subsequent babies.
4. Preoperative Blood Typing:
-
Primary Use: Performed before surgical procedures to ensure compatible blood is available in case transfusion is needed during or after surgery.
-
How it helps: Prepares surgical teams for emergencies by having the right blood type ready, ensuring that if unexpected bleeding occurs, life-saving transfusions can be given immediately without dangerous delays.
5. Blood Donor Screening:
-
Primary Use: Used to type blood donated at blood banks and donation centers, ensuring proper labeling and compatibility for future transfusion recipients.
-
How it helps: Builds and organizes the safe blood supply that hospitals rely on, ensuring that every donated unit is correctly labeled and can be matched to patients who need it.
6. Paternity Testing and Forensic Applications:
-
Primary Use: Blood grouping can provide supportive evidence in paternity determination and forensic investigations, though DNA typing is now more common.
-
How it helps: Offers a reliable, cost-effective method for initial screening in legal and forensic contexts, helping to establish biological relationships or exclude suspects in criminal investigations.
7. Population and Anthropological Studies:
-
Primary Use: Used in research to study distribution of blood groups in different populations and ethnic groups.
-
How it helps: Contributes to our understanding of human migration patterns, genetic diversity, and evolutionary history, providing valuable insights for anthropologists and genetic researchers.
SECONDARY & SUPPORTIVE USES
1. Quality Control in Blood Banks: Used for regular quality assurance of blood typing reagents and procedures, ensuring the accuracy and reliability of all blood typing performed in the facility.
2. Teaching and Training: Essential for training medical students, laboratory technicians, and phlebotomists in blood typing techniques, building the skills of future healthcare professionals.
3. Research and Genetic Studies: Used in studies investigating associations between blood groups and disease susceptibility, potentially uncovering important links that could lead to new treatments or preventive strategies.
4. Emergency and Disaster Response: Stocked in emergency medical kits for rapid blood typing in mass casualty situations, enabling quick transfusion decisions when every minute counts.
5. Field and Remote Clinic Use: Portable kits enable blood typing in resource-limited settings without laboratory infrastructure, bringing essential diagnostic capability to underserved communities.
6. Veterinary Medicine: Adapted for blood typing in animals, particularly in veterinary transfusion medicine, helping veterinarians provide life-saving transfusions to pets and livestock.
7. Anthropological Research: Used in studies of human migration and population genetics, helping researchers trace the movements and relationships of ancient populations through blood group distribution patterns.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: Complete diagnostic kit for determining ABO blood group and Rh factor.
-
Common Names: Blood Typing Kit, ABO/Rh Kit, Blood Grouping Kit, Blood Typing Sera Kit, Blood Grouping Reagents.
-
Kit Components:
-
Anti-A Monoclonal Antibodies: Blue-colored reagent for detecting A antigen.
-
Anti-B Monoclonal Antibodies: Yellow-colored reagent for detecting B antigen.
-
Anti-D (Rh) Monoclonal Antibodies: Colorless or specific color reagent for detecting Rh factor.
-
Control Reagents: Negative controls for quality assurance.
-
Reaction Cards or Slides: Disposable cards or glass slides with marked wells.
-
Mixing Sticks: Disposable sticks for mixing blood and reagents.
-
Lancets: Sterile lancets for fingerstick blood collection.
-
Capillary Tubes or Pipettes: For blood sample collection and transfer.
-
Package Insert: Detailed instructions and interpretation guide.
-
-
Sample Type: Whole blood (fingerstick or venipuncture), serum, or red blood cell suspension.
-
Test Time: 2-5 minutes for slide method; 15-30 minutes for tube method.
-
Result Interpretation: Agglutination (clumping) indicates presence of corresponding antigen.
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Sensitivity: >99.9% for ABO and Rh typing when used correctly.
-
Specificity: >99.9% for ABO and Rh typing; minimal cross-reactivity.
-
Test Principle: Agglutination (clumping) of red blood cells occurs when corresponding antigen and antibody interact.
-
Reagent Type: Monoclonal antibodies (most common) or polyclonal sera.
-
Reaction Reading: Visible agglutination = positive; smooth suspension = negative.
-
Grading: Agglutination strength graded from 1+ (weak) to 4+ (strong).
-
Stability: Reagents stable for 12-24 months at 2-8°C; do not freeze.
-
Controls: Built-in positive and negative controls ensure test validity.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Kit Size: 10-100 tests per kit depending on manufacturer.
-
Reagent Vials: 5-10 mL dropper bottles with color-coded labels.
-
Reaction Cards: Laminated cards with pre-marked circles for Anti-A, Anti-B, Anti-D, and controls.
-
Packaging: Compact box containing all components; some kits include a carrying case.
-
Storage Requirements: 2-8°C refrigeration required for reagents; do not freeze.
-
Shelf Life: 12-24 months depending on manufacturer and storage conditions.
-
Disposal: Used cards, lancets, and pipettes disposed as biohazardous waste.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device requiring FDA 510(k) clearance; CE marked for IVD use.
-
Quality Standards: Manufactured under ISO 13485; meets CLSI and AABB standards.
-
Biohazard Precautions: All blood samples potentially infectious; follow universal precautions.
-
Chemical Safety: Reagents contain sodium azide as preservative; avoid contact with eyes and skin.
-
Disposal: Dispose of used materials as biohazardous waste; lancets in sharps container.
-
Latex-Free: All components latex-free.
-
Quality Control: Controls included for each test run; external controls recommended.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage Temperature: 2-8°C; do not freeze; protect from light.
-
Reagent Handling: Bring reagents to room temperature before use; do not use expired reagents.
-
Sample Handling: Test fresh blood within 24 hours; refrigerate if delayed.
-
Test Procedure (Slide Method):
-
Place one drop of Anti-A, Anti-B, and Anti-D in separate wells on the reaction card.
-
Add one drop of blood or cell suspension to each well.
-
Mix with separate sticks for each well.
-
Rock the card gently for 1-2 minutes.
-
Observe for agglutination; record results.
-
-
Interpretation:
-
Anti-A (+) only: Blood group A.
-
Anti-B (+) only: Blood group B.
-
Anti-A (+) and Anti-B (+): Blood group AB.
-
Anti-A (-) and Anti-B (-): Blood group O.
-
Anti-D (+): Rh positive.
-
Anti-D (-): Rh negative.
-
Expiration: Never use expired reagents; results invalid.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Determination of ABO blood group and Rh factor for transfusion compatibility, prenatal care, and clinical management.
-
Clinical Significance:
-
Type O: Universal red cell donor; can receive only type O blood.
-
Type A: Can receive type A or O blood.
-
Type B: Can receive type B or O blood.
-
Type AB: Universal plasma donor; can receive A, B, AB, or O blood (universal recipient).
-
Rh Positive: Can receive Rh+ or Rh- blood.
-
Rh Negative: Should receive only Rh- blood (except in emergencies).
-
-
Forward and Reverse Typing: Complete kits may include reagents for both forward (cell typing) and reverse (serum typing) methods for confirmation.
-
Limitations: Weak subgroups may cause weak or false-negative reactions; cold agglutinins may interfere; requires trained personnel for interpretation.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Infection Control: All blood samples potentially infectious; wear gloves and appropriate PPE.
-
Sharps Safety: Lancets are single-use; dispose immediately in sharps containers.
-
Reagent Safety: Avoid contact with eyes and skin; wash hands after handling.
-
Quality Control: Always run controls with each batch; invalid if controls fail.
-
Expiration: Never use expired reagents; accuracy compromised.
-
Storage: Store reagents at proper temperature; heat or freezing destroys activity.
-
Interpretation: Read immediately at specified time; delayed reading may cause false positives.
-
Confirmation: Forward typing should be confirmed with reverse typing when possible.
-
Patient Identification: Ensure correct patient identification before testing and recording results.
2. FIRST AID MEASURES
-
Needlestick/Sharps Injury: Wash area vigorously with soap and water; report immediately; follow institutional exposure protocol.
-
Blood Spill: Contain with absorbent material; disinfect area with 10% bleach solution.
-
Reagent Contact (Eyes): Flush eyes with copious water for 15 minutes; seek medical attention.
-
Reagent Ingestion: Contact poison control center; do not induce vomiting unless instructed.
3. FIRE FIGHTING MEASURES
-
Flammability: Reagents contain alcohol; plastic components combustible.
-
Extinguishing Media: Use water, foam, CO₂, or dry chemical as appropriate.
-
Sodium Azide Hazard: Reagents contain sodium azide; may react with lead/copper plumbing to form explosive azides; flush with copious water when disposing.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties