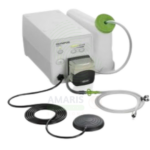
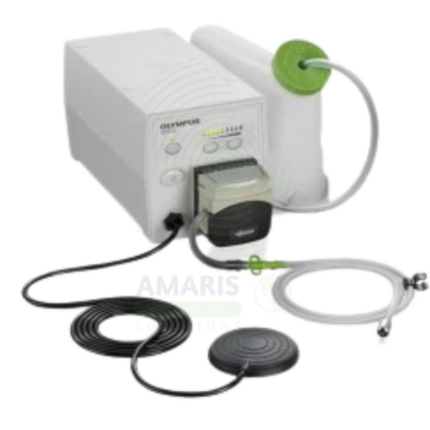
Active CO₂ Insufflator
WhatsApp Order
An Active CO₂ Insufflator delivers regulated carbon dioxide gas for luminal distension during endoscopic procedures. CO₂ is rapidly absorbed from the gastrointestinal tract, significantly reducing post-procedural pain, bloating, and abdominal distension compared to traditional air insufflation. With adjustable pressure and flow controls, automated pressure monitoring, and safety alarms, it provides stable visualization for diagnostic and therapeutic procedures while improving patient comfort and safety.
Description
Active CO₂ Insufflator
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Carbon Dioxide Insufflation for Endoscopic Procedures
-
Primary Use: Delivers controlled, regulated carbon dioxide gas into the lumen of the gastrointestinal tract during endoscopic procedures, creating a stable, distended view of the anatomy. Unlike room air insufflation, CO₂ is rapidly absorbed, significantly reducing post-procedural pain and discomfort.
-
How it helps: For the endoscopist and gastroenterologist, the CO₂ insufflator provides a stable, clear visual field while minimizing patient discomfort—the rapid absorption of CO₂ reduces abdominal distension and post-procedural bloating compared to traditional air insufflation. For the patient undergoing colonoscopy or upper endoscopy, CO₂ insufflation means less post-procedural pain, reduced bloating, and faster recovery.
2. Reduction of Post-Procedural Pain and Discomfort
-
Primary Use: CO₂ is rapidly absorbed from the gastrointestinal tract, reducing the volume of gas remaining after the procedure. This significantly decreases post-procedural pain, bloating, and abdominal distension compared to room air insufflation, improving patient satisfaction and recovery.
-
How it helps: For the patient, CO₂ insufflation transforms the post-procedure experience—replacing the uncomfortable bloating and cramping associated with air insufflation with minimal discomfort and faster return to normal activities. For the endoscopy unit, improved patient comfort leads to higher satisfaction scores and improved patient acceptance of screening procedures.
3. Precise Pressure and Flow Control
-
Primary Use: Provides adjustable pressure and flow settings to maintain consistent luminal distension throughout the procedure. The system monitors intra-luminal pressure and automatically adjusts gas delivery to maintain the preset pressure, preventing over-distension while ensuring adequate visualization.
-
How it helps: For the endoscopist, automated pressure regulation ensures consistent luminal distension throughout the procedure—maintaining optimal visualization without the need for manual adjustments. For the patient, this means a safer procedure with reduced risk of over-distension and barotrauma.
4. Enhanced Visualization for Therapeutic Procedures
-
Primary Use: Maintains stable luminal distension during complex therapeutic procedures including endoscopic submucosal dissection, endoscopic mucosal resection, and stent placement, providing the stable workspace needed for precise interventions.
-
How it helps: For the interventional endoscopist performing advanced resection procedures, stable CO₂ insufflation provides the consistent luminal distension needed to safely and effectively remove lesions—maintaining visualization throughout lengthy procedures. For the patient undergoing therapeutic endoscopy, stable insufflation enables precise, complete resection with minimal complications.
5. Safety Features for High-Risk Patients
-
Primary Use: Automated pressure monitoring and safety alarms prevent over-insufflation, reducing the risk of perforation and barotrauma. This is particularly important in patients with compromised bowel walls, such as those with inflammatory bowel disease or following recent surgery.
-
How it helps: For the endoscopist, integrated safety features provide peace of mind during procedures on high-risk patients—automatically limiting pressure to prevent over-distension and alerting the operator to potential complications. For the patient with inflammatory bowel disease, prior abdominal surgery, or other risk factors, these safety features reduce the risk of perforation and other complications.
SECONDARY & SUPPORTIVE USES
1. Endoscopic Retrograde Cholangiopancreatography: CO₂ insufflation during ERCP procedures to visualize the biliary and pancreatic ducts with reduced post-procedural pain.
2. Enteroscopy: Deep small bowel examination with CO₂ insufflation for improved patient comfort during lengthy procedures.
3. Barotrauma Prevention: Automated pressure regulation prevents excessive luminal distension that could lead to perforation.
4. Screening Colonoscopy: Enhanced patient comfort improves acceptance and compliance with colorectal cancer screening programs.
5. Sedation Reduction: Improved patient tolerance may reduce sedation requirements in some patients.
6. Pediatric Endoscopy: CO₂ insufflation is particularly beneficial in children due to reduced post-procedural pain and faster recovery.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: A medical device that delivers regulated carbon dioxide gas for insufflation during endoscopic procedures.
-
Designation: Active CO₂ Insufflator, CO₂ Insufflator, Endoscopic CO₂ Insufflator, Carbon Dioxide Insufflator.
-
Key Components:
-
CO₂ Source Connection: Standard medical gas connection for CO₂ supply.
-
Pressure Regulator: Controls gas delivery pressure.
-
Flow Controller: Adjustable flow rates for different procedures.
-
Pressure Monitoring: Real-time monitoring of intra-luminal pressure.
-
Safety Alarms: Visual and audible alarms for pressure limits.
-
Tubing Set: Single-use sterile tubing for patient connection.
-
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Gas Source: Medical grade CO₂ (99.5% purity).
-
Pressure Range: Adjustable up to 20-40 mmHg depending on procedure.
-
Flow Rate: Adjustable flow rates for different applications.
-
Safety Features: Over-pressure protection, automatic shut-off, pressure alarms.
-
Absorption Rate: CO₂ is absorbed 100-150 times faster than room air.
-
Power: AC power with battery backup options.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Construction: Compact, mobile unit with locking casters.
-
Controls: Digital interface with touchscreen or membrane controls.
-
Tubing: Single-use, sterile tubing sets for each patient.
-
Portability: Cart-mounted or tabletop configurations.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device regulated by FDA.
-
Electrical Safety: Compliant with IEC 60601-1.
-
Gas Safety: Certified for use with medical grade CO₂.
-
Pressure Safety: Over-pressure protection and safety alarms.
-
Single-Use Components: Tubing sets are sterile, single-use, disposable.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Stored in the procedure room or equipment cart.
-
Cleaning: Wipe external surfaces with hospital-grade disinfectants.
-
Maintenance: Regular calibration and preventive maintenance.
-
Gas Supply: Ensure adequate CO₂ supply before procedure.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Gas insufflation for gastrointestinal endoscopic procedures.
-
Clinical Role: Essential for modern endoscopy, improving patient comfort and safety.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Pressure Monitoring: Monitor pressure settings to prevent over-insufflation.
-
Gas Supply: Ensure adequate CO₂ supply before starting procedure.
-
Tubing Integrity: Ensure tubing connections are secure; use sterile, single-use tubing for each patient.
-
Alarm Response: Never ignore pressure alarms; respond immediately to prevent complications.
-
Patient Selection: Use with caution in patients with known CO₂ retention or severe respiratory compromise.
2. FIRST AID MEASURES
-
Over-Insufflation: If over-insufflation occurs, reduce or stop gas flow; assess patient.
-
Equipment Failure: If the insufflator fails, manual insufflation is available.
-
Gas Leak: If CO₂ leak is detected, ventilate area; ensure proper ventilation.
3. FIRE FIGHTING MEASURES
-
Flammability: CO₂ is non-flammable; plastic components are combustible.
-
Extinguishing Media: Use water, foam, or CO₂ as appropriate for surrounding materials.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties