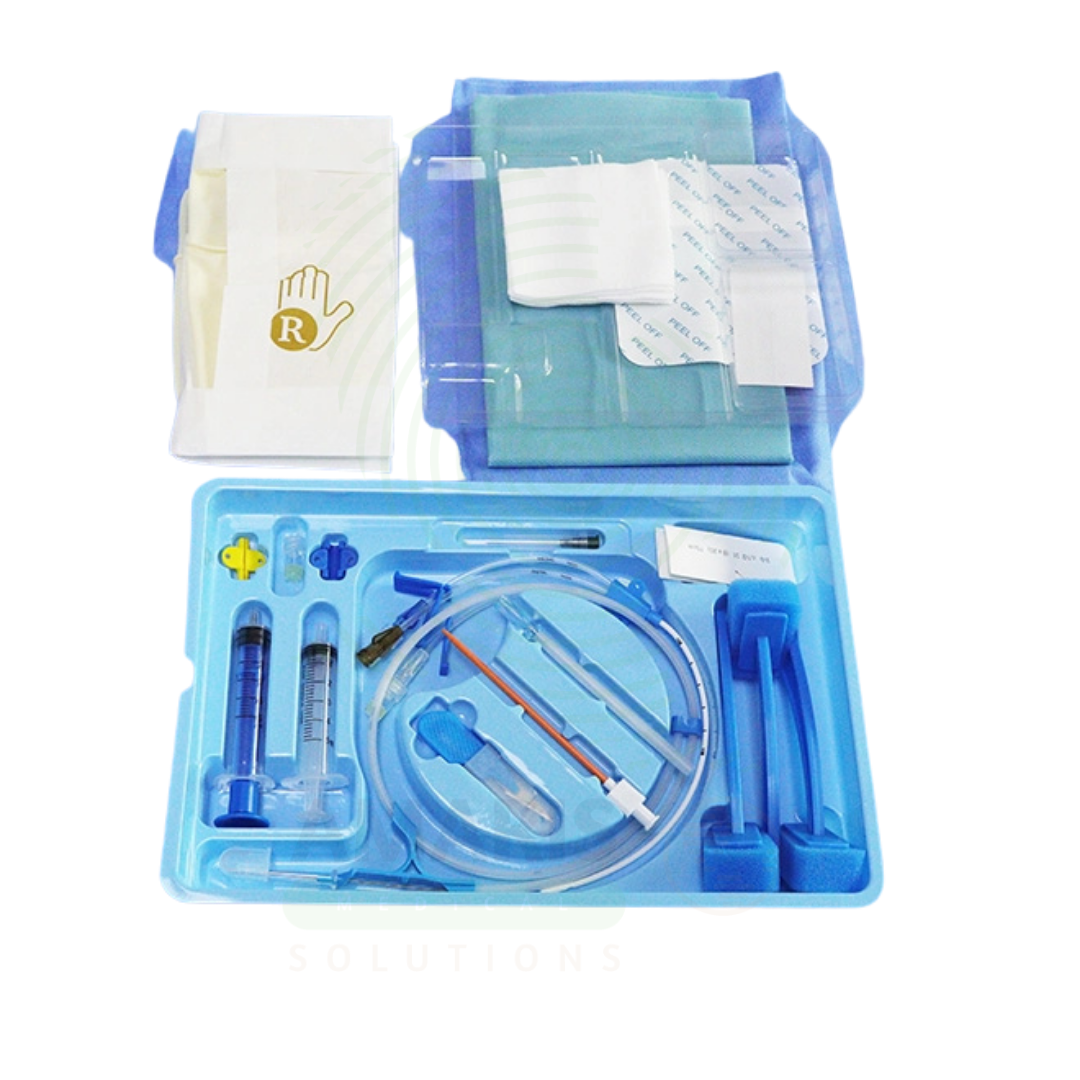
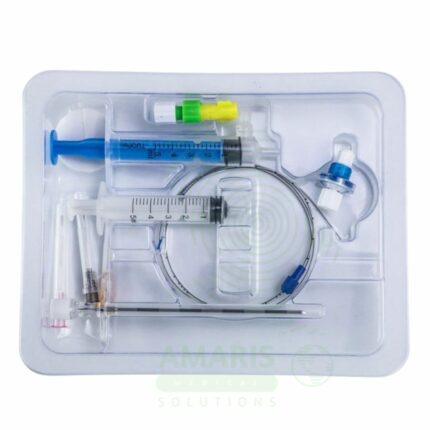
CVC Mini-kit
WhatsApp Order
A CVC Mini-kit is a sterile, single-use convenience kit containing all components necessary for central venous catheter insertion site preparation. Including sterile drapes, antiseptic solutions, gauze, syringes, needles, and dressings, it ensures consistent aseptic technique and reduces the risk of catheter-related bloodstream infections. Essential for intensive care, anesthesia, and interventional radiology.
Description
CVC Mini-kit
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Central Venous Catheter Insertion Preparation
-
Primary Use: Provides a complete, sterile set of components for preparing the patient and site for central venous catheter insertion. The mini-kit includes sterile drapes, antiseptic solutions, gauze, and other supplies needed for establishing a sterile field and preparing the insertion site.
-
How it helps: For the intensivist, anesthesiologist, and interventional radiologist, the CVC mini-kit streamlines the preparation process for central line placement—ensuring that all necessary sterile components are readily available and organized. For the patient, this means consistent adherence to sterile technique, reducing the risk of catheter-related bloodstream infections.
2. Site Preparation and Skin Antisepsis
-
Primary Use: Contains antiseptic solutions (chlorhexidine, povidone-iodine) and applicators for thorough skin preparation before CVC insertion, reducing the risk of infection at the insertion site.
-
How it helps: For the clinician performing central line placement, the included antiseptics ensure proper skin preparation according to infection prevention guidelines. For the patient, meticulous site preparation significantly reduces the risk of catheter-related bloodstream infections.
3. Sterile Field Establishment
-
Primary Use: Includes fenestrated and non-fenestrated sterile drapes for creating a sterile field around the insertion site, isolating the area from non-sterile surfaces and equipment.
-
How it helps: For the procedural team, sterile drapes provide a defined sterile field that maintains the aseptic environment throughout the procedure. For the patient, this reduces the risk of contamination during catheter insertion.
4. Local Anesthesia Preparation
-
Primary Use: Provides syringes and needles for drawing up and administering local anesthetic, ensuring that sterile equipment is available for infiltration of the insertion site.
-
How it helps: For the clinician, the included syringes and needles simplify the preparation of local anesthetic—reducing setup time and ensuring sterility. For the patient, this ensures that local anesthesia is administered with sterile equipment.
5. Wound Management and Dressing Application
-
Primary Use: Includes gauze, sterile dressings, and transparent film dressings for covering the insertion site after catheter placement, maintaining sterility and allowing for visual monitoring.
-
How it helps: For the post-procedure care team, the included dressings ensure that the insertion site is properly covered and protected after catheter placement. For the patient, proper dressing reduces infection risk and allows for early detection of site complications.
SECONDARY & SUPPORTIVE USES
1. Arterial Line Insertion: Can be used for preparing arterial line placement procedures.
2. Peripheral Line Insertion: Provides sterile supplies for difficult peripheral IV access procedures.
3. PICC Line Insertion: Supplies for peripherally inserted central catheter placement.
4. Dialysis Catheter Insertion: Used for preparing insertion site for dialysis catheters.
5. Biopsy Procedures: Provides sterile field supplies for guided biopsy procedures.
6. Emergency Line Placement: Ready-to-use kit for urgent vascular access procedures.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: A sterile, single-use convenience kit for central venous catheter insertion preparation.
-
Designation: CVC Mini-kit, Central Line Insertion Kit, Central Venous Catheter Kit, CVC Insertion Tray.
-
Key Components:
-
Sterile Drapes: Fenestrated and non-fenestrated drapes.
-
Antiseptic Solutions: Chlorhexidine, povidone-iodine, or alcohol swabs.
-
Applicators: Sponge applicators for antiseptic application.
-
Gauze Sponges: Various sizes for site preparation.
-
Syringes: 5-10 mL syringes for local anesthetic.
-
Needles: Hypodermic needles for anesthetic draw-up and infiltration.
-
Sterile Gloves: One or more pairs of sterile gloves.
-
Dressings: Transparent film dressings and gauze.
-
Tape: Adhesive tape for securing dressings.
-
Disposal Bag: For safe disposal of used materials.
-
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Sterility: Gamma irradiated or ethylene oxide sterilized.
-
Packaging: Sealed, peel-open sterile packaging.
-
Components: Pre-assembled in procedure-specific layout.
-
Antiseptics: Chlorhexidine or povidone-iodine based.
-
Gloves: Powder-free, latex-free options available.
-
Latex-Free: Available in latex-free formulations.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Tray Configuration: Components organized in sequential layout.
-
Packaging: Easy-open sterile peel pouch.
-
Portability: Compact for storage in procedure rooms.
-
Disposal: Entire unit disposed as medical waste after use.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device regulated by FDA.
-
Sterility Assurance: Validated sterilization process.
-
Latex-Free Options: Available for patients and staff with latex sensitivity.
-
Biocompatibility: Materials meet biocompatibility requirements.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in cool, dry locations; protect from moisture.
-
Sterility Maintenance: Do not use it if the package is opened, damaged, or wet.
-
Expiration: Check expiration date before use; do not use after expiration.
-
Single-Use Only: Intended for single patient use only; do not resterilize or reuse.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Sterile preparation for central venous catheter insertion.
-
Clinical Role: Essential for maintaining aseptic technique during vascular access procedures.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Sterile Technique: Maintain sterile technique throughout the procedure.
-
Antiseptic Use: Allow antiseptic to dry before catheter insertion.
-
Proper Disposal: Dispose of all used components in provided disposal bag or appropriate waste container.
-
Drape Placement: Ensure drapes are properly positioned to maintain sterile fields.
-
Glove Selection: Use appropriate glove size for proper fit and dexterity.
2. FIRST AID MEASURES
-
Antiseptic Reaction: If a patient shows signs of allergic reaction to antiseptic, discontinue use; seek medical attention.
-
Contamination: If the sterile field is contaminated during the procedure, discard and use a new kit.
-
Needlestick: If needlestick occurs, follow institutional sharps injury protocol.
3. FIRE FIGHTING MEASURES
-
Flammability: Drapes, gauze, and paper components are combustible; antiseptic solutions may be flammable.
-
Extinguishing Media: Use water, foam, or CO₂ as appropriate for surrounding materials.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties