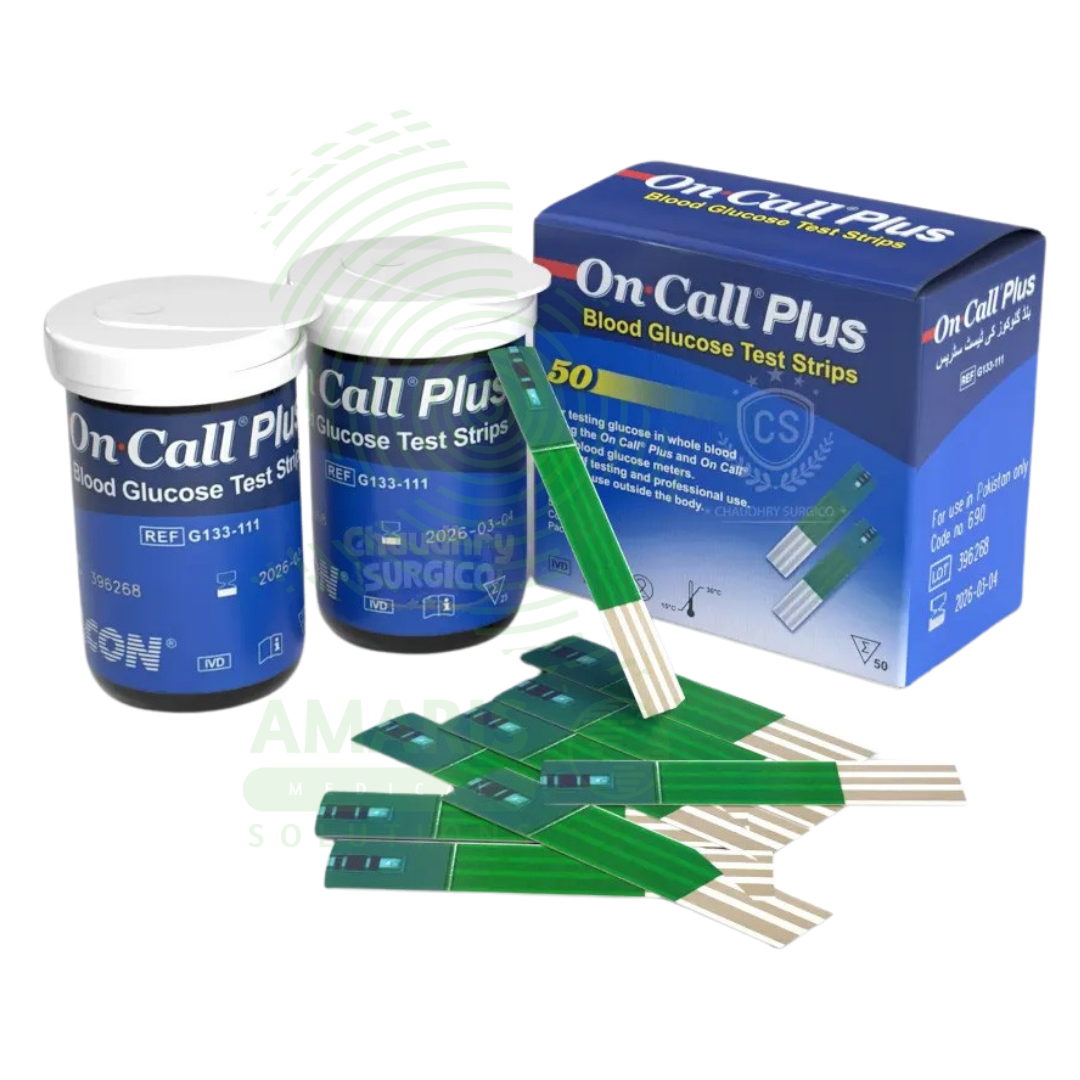
On Call Blood Sugar Strips
WhatsApp Order
On Call Blood Sugar Strips are single-use, disposable electrochemical test strips (ISO 15197:2013 compliant) designed exclusively for use with On Call glucose meters to measure capillary blood glucose from 0.5-1.0 µL samples in 5-7 seconds. Each strip contains glucose oxidase or glucose dehydrogenase reagents that generate electrical current proportional to glucose concentration (20-600 mg/dL range). Packaging options include multi-strip vials (25-100 strips) with desiccant for daily use or individually foil-wrapped strips for travel. Strips feature no-coding automatic calibration, hematocrit compensation (20-60%), and interference resistance. Primary clinical applications include daily blood glucose testing for Type 1, Type 2, and gestational diabetes management, hypoglycemia and hyperglycemia detection, pre- and post-meal glucose assessment, and hospital point-of-care testing. Critical safety precautions include single-use only, immediate vial closure after strip removal to prevent humidity damage, storage at 2-30°C away from bathrooms and temperature extremes, never using expired strips, proper hand hygiene before testing, and disposal in sharps containers. Essential consumable for diabetes self-management when used with On Call meters.
Description
On Call Blood Sugar Strips
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Daily Blood Glucose Testing for Diabetes Management:
-
Primary Use: Single-use disposable strips work exclusively with On Call glucose meters to measure capillary blood glucose levels multiple times daily, enabling individuals with Type 1, Type 2, or gestational diabetes to obtain accurate readings essential for insulin dosing, dietary adjustments, and lifestyle modifications.
-
How it helps: Provides the essential consumable that makes daily glucose monitoring possible, giving patients the reliable, accurate readings they need to make confident decisions about their diabetes management moment by moment.
2. Hypoglycemia Detection and Confirmation:
-
Primary Use: Enables rapid confirmation of suspected low blood sugar (hypoglycemia) when symptoms such as shakiness, sweating, confusion, or weakness occur, allowing immediate corrective action to prevent progression to unconsciousness or seizures.
-
How it helps: Serves as a critical safety tool that takes the guesswork out of scary symptoms, allowing patients to quickly confirm whether they are experiencing dangerous lows and take immediate action before the situation becomes a medical emergency.
3. Hyperglycemia Identification and Monitoring:
-
Primary Use: Facilitates detection of high blood sugar (hyperglycemia) in patients experiencing symptoms such as increased thirst, frequent urination, or blurred vision, guiding corrective insulin doses and preventing diabetic ketoacidosis (DKA).
-
How it helps: Empowers patients to catch high blood sugar early before it spirals into a serious complication, giving them the information they need to take corrective action and avoid hospitalization.
4. Pre-Meal and Post-Meal Glucose Assessment:
-
Primary Use: Allows patients to test glucose levels before and after meals to understand how different foods affect their blood sugar, enabling informed dietary choices and precise mealtime insulin dosing.
-
How it helps: Transforms eating from a source of anxiety into an opportunity for learning, helping patients discover which foods work best for their body and how to dose insulin accurately for better control after meals.
5. Gestational Diabetes Management:
-
Primary Use: Essential for pregnant women with gestational diabetes to perform prescribed fasting and post-meal testing, ensuring tight glycemic control critical for preventing fetal macrosomia, neonatal hypoglycemia, and delivery complications.
-
How it helps: Protects both mother and baby during pregnancy by providing the accurate readings needed to keep blood sugar tightly controlled, preventing complications that could affect the baby’s health and the safety of delivery.
6. Hospital and Clinical Point-of-Care Testing:
-
Primary Use: Used by healthcare providers in hospitals, clinics, and emergency departments with On Call professional meters for rapid bedside glucose assessment of inpatients and outpatients.
-
How it helps: Delivers instant results at the bedside, enabling clinicians to make immediate treatment decisions without waiting for laboratory confirmation, which is essential for managing critically ill patients and busy clinical environments.
7. Long-Term Glycemic Trend Monitoring:
-
Primary Use: Serial testing with On Call strips provides data for calculating average glucose levels, time-in-range metrics, and estimated HbA1c, enabling comprehensive assessment of diabetes management effectiveness.
-
How it helps: Transforms individual readings into a powerful long-term health picture, showing doctors and patients how well their diabetes management plan is working over weeks and months and guiding strategic adjustments.
SECONDARY & SUPPORTIVE USES
1. Medication Adjustment Monitoring: Used to track glucose responses when starting or changing doses of diabetes medications, including insulin, metformin, sulfonylureas, and GLP-1 agonists, helping optimize treatment regimens.
2. Exercise-Induced Glucose Response Testing: Enables patients to test before, during, and after physical activity to understand exercise effects on blood sugar and prevent exercise-related hypoglycemia.
3. Sick-Day Management: Critical for more frequent testing during illness when glucose levels may fluctuate unpredictably, guiding insulin adjustments and preventing DKA.
4. Fasting Glucose Monitoring: Used to assess fasting glucose levels for diabetes diagnosis and ongoing management of morning hyperglycemia.
5. Post-Bariatric Surgery Monitoring: Helps track glycemic status in patients who may experience diabetes remission following bariatric procedures, ensuring continued metabolic health.
6. Prediabetes and Insulin Resistance Assessment: Used in diagnostic protocols and research settings to evaluate glucose patterns in at-risk individuals.
7. Travel and Lifestyle Management: Portable strips enable consistent monitoring during travel, work, and daily activities, maintaining diabetes control across varied environments.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: Disposable electrochemical test strips for quantitative measurement of glucose in capillary blood.
-
Common Names: On Call Test Strips, On Call Glucose Strips, Blood Sugar Test Strips, Diabetic Test Strips.
-
Compatibility: Specifically designed for use with On Call brand glucose meters only; not interchangeable with other meter brands.
-
Components:
-
Strip Base: Plastic substrate with printed electrodes and reagents.
-
Sample Tip: Capillary fill channel at strip end for blood application.
-
Contact Bars: Metal or conductive area that inserts into meter port.
-
Reagent Layer: Contains glucose oxidase or glucose dehydrogenase enzyme, mediators, and stabilizers.
-
Packaging: Foil-wrapped vial or individual foil pouches protecting strips from moisture and light.
-
-
Sample Type: Fresh capillary whole blood from fingertip (standard).
-
Sample Volume: 0.5-1.0 µL typical.
-
Measurement Range: 20-600 mg/dL (1.1-33.3 mmol/L).
-
Test Time: 5-7 seconds.
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Accuracy Standards: Meets ISO 15197:2013 requirements; ≥95% of results within ±15 mg/dL (<100 mg/dL) or ±15% (≥100 mg/dL) of laboratory reference.
-
Measurement Principle: Electrochemical biosensor; glucose in blood sample reacts with reagents generating electrical current proportional to glucose concentration.
-
Enzyme Type: Glucose oxidase or glucose dehydrogenase (specific to On Call formulation).
-
Hematocrit Range: Compensated for hematocrit 20-60% to minimize interference.
-
Interference Resistance: Formulated to minimize effects of common interfering substances (acetaminophen, ascorbic acid, uric acid).
-
Coding: No-coding system; strip automatically calibrated when inserted.
-
Storage Stability: Stable until expiration date when stored properly.
-
In-Use Stability: Must be used within specified time after removal from vial (typically 3-5 minutes).
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Strip Dimensions: Approximately 3-4 cm length × 0.5 cm width.
-
Strip Color: Typically white or light-colored with colored contact bars.
-
Packaging Options:
-
Vial Packaging: 25-100 strips per vial with desiccant; economical for daily use.
-
Individual Foil Wrapping: Single strips individually sealed; convenient for travel or occasional use.
-
-
Vial Design: Child-resistant cap; moisture-proof seal; desiccant included.
-
Shelf Life: 18-24 months from manufacture date when unopened.
-
In-Vial Life: 3-6 months after opening if vial properly sealed.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device requiring FDA 510(k) clearance; used only with cleared On Call meters.
-
Clinical Standards: Meets ISO 15197:2013 accuracy requirements for blood glucose monitoring systems.
-
Quality Management: Manufactured under ISO 13485 certified processes.
-
Sterility: Strips are not sterile but manufactured under clean conditions; single-patient use only.
-
Biocompatibility: Materials meet ISO 10993 for blood contact; non-toxic, non-irritating.
-
Latex-Free: All components latex-free.
-
Packaging Integrity: Foil and vial packaging maintain strip integrity until opened.
-
Disposal: Used strips considered biohazardous waste; dispose in sharps containers.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage Temperature: Store unopened vials at 2-30°C (36-86°F). Avoid freezing and extreme heat.
-
Humidity Protection: Store in original sealed vial; do not transfer to other containers. Keep away from bathrooms, sinks, and humid areas.
-
Light Protection: Store in original opaque packaging; protect from direct sunlight.
-
In-Use Storage: After opening, keep the vial tightly closed; remove only one strip at a time. Do not touch strip contact bars.
-
Expiration: Never use strips beyond expiration date printed on vial. Discard opened vial 3-6 months after first opening (per manufacturer).
-
Visual Inspection: Do not use strips that appear damaged, discolored, or if the vial seal is broken.
-
Temperature Excursions: Do not use strips exposed to temperatures outside recommended range; accuracy compromised.
-
Transport: When traveling, keep strips in original vial; avoid temperature extremes in cars or luggage.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Single-use consumable for quantitative blood glucose measurement when used with On Call meters, essential for daily diabetes self-management.
-
Testing Frequency Guidelines (ADA):
-
Type 1 Diabetes: 4-10 strips daily (before meals, post-meals, before bed, before exercise, when hypoglycemia is suspected).
-
Type 2 Diabetes (Insulin-Treated): 2-8 strips daily as above.
-
Type 2 Diabetes (Non-Insulin): 1-7 strips weekly depending on glycemic control.
-
Gestational Diabetes: 4-7 strips daily (fasting and post-meal testing).
-
-
Proper Testing Technique:
-
Wash hands with soap and water; dry thoroughly.
-
Remove one strip from vial; close vial immediately.
-
Insert strip into meter; meter turns on automatically.
-
Prick fingertip with lancet; obtain small blood drop.
-
Touch strip sample tip to blood drop; blood is drawn in automatically.
-
Wait 5-7 seconds for the result.
-
Remove and discard used strips in a sharps container.
-
Quality Control: Use On Call control solution to verify strip and meter accuracy with each new vial and periodically as needed.
-
Limitations: For use with On Call meters only; not compatible with other brands. Results for capillary blood; venous or arterial samples may differ.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Single-Use Only: Strips are for single use only; never reuse a strip. Reuse causes inaccurate results and infection risk.
-
Hand Hygiene: Always wash hands before testing; food residue, sweat, or lotion on fingers causes inaccurate results.
-
Vial Closure: Close vial immediately after removing strip; humidity exposure degrades strips within hours.
-
Expiration Dates: Never use expired strips; accuracy cannot be guaranteed and may lead to incorrect insulin dosing.
-
Temperature Exposure: Never store strips in bathroom (humidity) or car (extreme temperatures). Heat and humidity permanently damage strips.
-
Blood Application: Apply blood only to designated sample tip; do not apply blood to top of strip or touch strip to finger repeatedly.
-
Insufficient Sample: If meter displays error for insufficient blood, discard strip and use new strip; do not attempt to add more blood.
-
Control Solution Testing: Test with control solution when opening a new vial, if meter is dropped, or if results seem inconsistent with how you feel.
-
Proper Disposal: Dispose of used strips in FDA-cleared sharps container; never discard loose in household trash.
-
Keep Away from Children: Strips are small and pose choking hazard; desiccant in vial may be harmful if ingested.
2. FIRST AID MEASURES
-
Strip Ingestion (Child): If a child swallows a test strip, contact the poison control center. Strips are generally non-toxic but may cause choking.
-
Desiccant Ingestion: If desiccant packet from vial is swallowed, contact poison control; may cause gastrointestinal irritation.
-
Blood Exposure: If another person contacts a used strip with blood, follow bloodborne pathogen exposure protocol.
-
Eye Contact with Blood: If blood splashes into the eye, flush with copious water; seek medical attention.
3. FIRE FIGHTING MEASURES
-
Flammability: Plastic strips and foil packaging are combustible.
-
Extinguishing Media: Use water, foam, CO₂, or dry chemical as appropriate for surrounding fire.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties