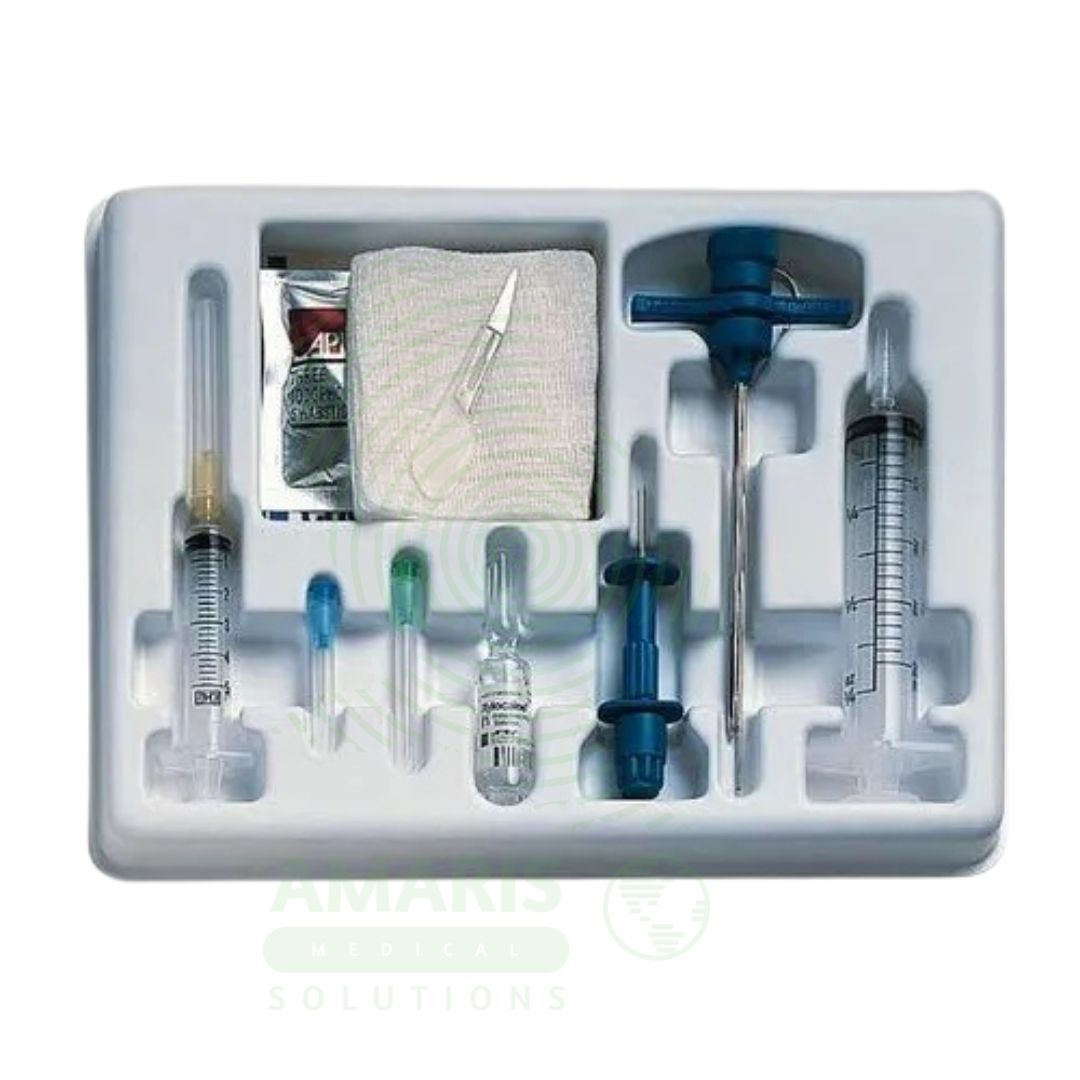
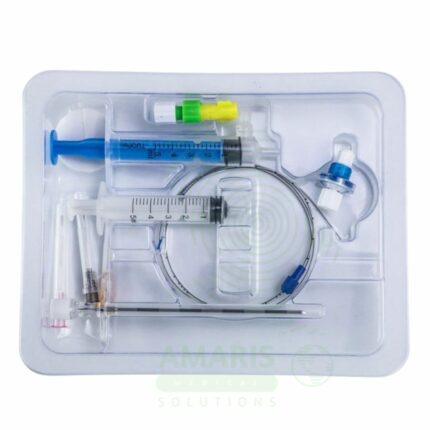
Bone Marrow Puncture Kit
WhatsApp Order
A Bone Marrow Puncture Kit is a sterile, single-use procedure tray containing all components necessary for bone marrow aspiration and core biopsy. Used for diagnosing hematologic malignancies, evaluating bone marrow function, and staging metastatic disease, the kit includes aspiration and biopsy needles, syringes, glass slides, fixatives, and specimen containers. Proper technique includes verifying coagulation status, administering local anesthesia, limiting aspiration volume, and preparing smears immediately. Essential for hematology-oncology practice, the kit enables the collection of specimens for histopathology, flow cytometry, cytogenetics, and molecular testing that guide diagnosis and treatment of blood disorders.
Description
Bone Marrow Puncture Kit
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Bone Marrow Aspiration for Hematologic Diagnosis
-
Primary Use: Provides sterile instruments for obtaining bone marrow aspirate from the iliac crest, sternum, or other sites for diagnostic evaluation of hematologic conditions including leukemia, lymphoma, myeloma, myelodysplastic syndromes, aplastic anemia, and metastatic disease. The kit contains specialized needles, syringes, and slides for collecting and preparing specimens.
-
How it helps: For the hematologist and oncologist, the bone marrow puncture kit provides the tools needed to obtain the tissue that reveals the health of the blood-forming system—allowing for definitive diagnosis of blood cancers, assessment of marrow function in cytopenias, and staging of lymphoproliferative disorders. For the patient with unexplained anemia, suspected leukemia, or abnormal blood counts, a bone marrow biopsy provides the definitive answers needed to guide treatment and prognosis.
2. Bone Marrow Core Biopsy for Histopathological Examination
-
Primary Use: Contains trephine biopsy needles for obtaining a solid core of bone marrow tissue, preserving the architectural relationship of cells within the marrow space. This provides critical information about cellularity, fibrosis, infiltration, and overall marrow structure that cannot be obtained from aspirate alone.
-
How it helps: For the pathologist and hematologist, the core biopsy specimen provides the architectural context essential for accurate diagnosis—revealing the degree of cellularity, presence of fibrosis, pattern of infiltration by malignant cells, and overall marrow architecture. For the patient with suspected myelofibrosis, myelodysplasia, or metastatic cancer, the core biopsy provides the structural information that completes the diagnostic picture.
3. Preparation of Bone Marrow Smears and Slides
-
Primary Use: Includes glass slides, spreaders, and fixatives for preparing bone marrow aspirate smears immediately after collection. Proper smear preparation is essential for cellular morphology evaluation, differential cell counts, and assessment of blast percentage in acute leukemias.
-
How it helps: For the laboratory technician and pathologist, properly prepared bone marrow smears are the foundation of accurate cytologic evaluation—preserving cell morphology, enabling differential counts, and allowing identification of abnormal cell populations. For the patient, high-quality smears mean that a single specimen can yield complete diagnostic information without the need for repeat procedures.
4. Flow Cytometry and Molecular Testing Specimen Collection
-
Primary Use: Provides sterile syringes, needles, and collection tubes with anticoagulants (typically heparin or EDTA) for collecting bone marrow aspirate for flow cytometry, cytogenetics, and molecular testing. These specialized studies are essential for immunophenotyping, detecting chromosomal abnormalities, and identifying genetic mutations in hematologic malignancies.
-
How it helps: For the hematopathologist and molecular diagnostic specialist, properly collected and preserved bone marrow specimens enable advanced testing that defines the specific subtype of leukemia or lymphoma, identifies targetable mutations, and guides precision therapy selection. For the patient, this means their treatment can be tailored to the specific genetic profile of their disease, improving outcomes and reducing unnecessary side effects.
5. Measurement of Bone Marrow Cellularity and Iron Stores
-
Primary Use: Provides specimens for evaluating marrow cellularity (normocellular, hypercellular, hypocellular) and assessing iron stores using special stains. This is critical for diagnosing disorders such as aplastic anemia, myelodysplasia, and iron deficiency or overload states.
-
How it helps: For the hematologist evaluating a patient with unexplained cytopenias, the bone marrow specimen provides quantitative and qualitative information about the marrow environment—revealing whether the problem is due to production failure, ineffective hematopoiesis, or peripheral destruction. For the patient with anemia, these findings guide the distinction between bone marrow failure syndromes and nutritional deficiencies.
SECONDARY & SUPPORTIVE USES
1. Staging of Lymphoma and Solid Tumors: Used to obtain bone marrow specimens for detecting metastatic involvement in patients with lymphoma, neuroblastoma, small cell lung cancer, breast cancer, and prostate cancer.
2. Assessment of Treatment Response: Serial bone marrow examinations are used to monitor response to chemotherapy in patients with acute leukemia, documenting remission status and detecting early relapse.
3. Evaluation of Unexplained Cytopenias: Provides diagnostic tissue for patients with unexplained anemia, thrombocytopenia, or leukopenia where peripheral blood evaluation is inconclusive.
4. Investigation of Fever of Unknown Origin: Bone marrow culture and histology may identify infectious organisms (e.g., tuberculosis, histoplasmosis, leishmaniasis) or infiltrative disorders in patients with FUO.
5. Research and Clinical Trials: Used to obtain bone marrow specimens for research protocols investigating new treatments for hematologic disorders and for correlative science studies.
6. Pre-Transplant Evaluation: Bone marrow examination is part of the pre-transplant workup for patients undergoing stem cell transplantation.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: A sterile, single-use procedure tray containing all components necessary for bone marrow aspiration and biopsy.
-
Designation: Bone Marrow Puncture Kit, Bone Marrow Biopsy Kit, Bone Marrow Aspiration Tray, Bone Marrow Procedure Kit, Jamshidi Needle Kit.
-
Key Components:
-
Aspiration Needle: Sharp, stylet needle (typically 11-16 gauge) for aspirating liquid marrow; may be Jamshidi-type or other design.
-
Trephine Biopsy Needle: Larger bore needle (typically 8-11 gauge) with cutting tip for obtaining core tissue specimen.
-
Disposable Syringes: Various sizes (10-20 mL) for aspiration and local anesthetic injection.
-
Glass Slides: Frosted-end slides for preparing marrow smears; typically 10-20 slides per kit.
-
Slide Spreader: Device or additional slide for creating monolayer smears.
-
Fixative: Methanol or other fixative for immediate fixation of slides.
-
Specimen Containers: Sterile containers with formalin for core biopsy specimen preservation.
-
Collection Tubes: Vacutainer tubes with anticoagulant (EDTA, heparin) for flow cytometry and molecular studies.
-
Local Anesthetic Syringes: Syringes with needles for lidocaine administration.
-
Sterile Drapes and Gauze: For maintaining sterile fields during procedure.
-
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Needle Gauge: Aspiration needles typically 11-16 gauge; biopsy needles 8-11 gauge.
-
Needle Length: Available in various lengths for different patient body habitus; typically 60-100 mm.
-
Stylet Design: Removable stylet prevents tissue coring during insertion.
-
Luer Lock Connections: Secure syringe connections for aspiration and injection.
-
Sterility: Gamma irradiated or ethylene oxide sterilized; pyrogen tested.
-
Latex-Free: Manufactured without natural rubber latex.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Tray Configuration: All components organized in sequential, procedure-specific layout.
-
Packaging: Sealed, peel-open sterile packaging; inner tray for sterile field presentation.
-
Disposal: Entire unit disposed as regulated medical waste after single use.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device regulated by FDA.
-
Sterility Assurance: Sterility assurance level validated.
-
Biocompatibility: Materials meet biological safety testing requirements.
-
Pyrogen-Free: Manufactured to be pyrogen-free.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in cool, dry locations; protect from moisture.
-
Sterility Maintenance: Do not use it if the package is opened, damaged, or wet.
-
Expiration: Check expiration date before use; do not use after expiration.
-
Single-Use Only: Devices are intended for single patient use only; do not resterilize or reuse.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Bone marrow aspiration and biopsy for diagnosis of hematologic disorders, staging of malignancies, and evaluation of bone marrow function.
-
Clinical Role: Essential procedure tray for hematology-oncology services, used in outpatient clinics, inpatient units, and operating rooms.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Coagulation Status: Verify normal coagulation parameters before procedure; platelet count should be adequate (typically >50,000/µL) to reduce bleeding risk.
-
Informed Consent: Obtain informed consent; explain procedure, risks, and alternatives.
-
Aseptic Technique: Strict aseptic technique is mandatory to prevent introduction of pathogens into bone marrow.
-
Site Selection: Select appropriate aspiration site based on patient anatomy and clinical indication; common sites include posterior iliac crest, anterior iliac crest, and sternum (sternal aspiration only).
-
Local Anesthesia: Administer adequate local anesthesia; may include deep periosteal infiltration.
-
Aspiration Volume: Limit aspiration volume to 0.2-0.5 mL to avoid excessive hemodilution with peripheral blood.
-
Smear Preparation: Prepare smears immediately after aspiration to prevent clotting and preserve cellular morphology.
-
Specimen Labeling: Label all specimens with patient identification, site, and collection time.
-
Post-Procedure Care: Apply pressure to site; monitor for bleeding; instruct patient to avoid strenuous activity for 24-48 hours.
2. FIRST AID MEASURES
-
Bleeding: If persistent bleeding occurs, apply firm pressure; if bleeding continues, seek medical attention.
-
Pain: Manage post-procedure pain with analgesics as prescribed.
-
Infection: Signs of infection (fever, increasing pain, erythema, drainage) require prompt medical evaluation.
-
Needlestick Injury: If injury occurs with a contaminated needle, follow institutional bloodborne pathogen exposure protocol.
3. FIRE FIGHTING MEASURES
-
Flammability: Plastic components are combustible; formalin fixative may be flammable.
-
Extinguishing Media: Use water, foam, or CO₂ as appropriate for surrounding materials.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties