Hepatitis B Test Kit
WhatsApp Order
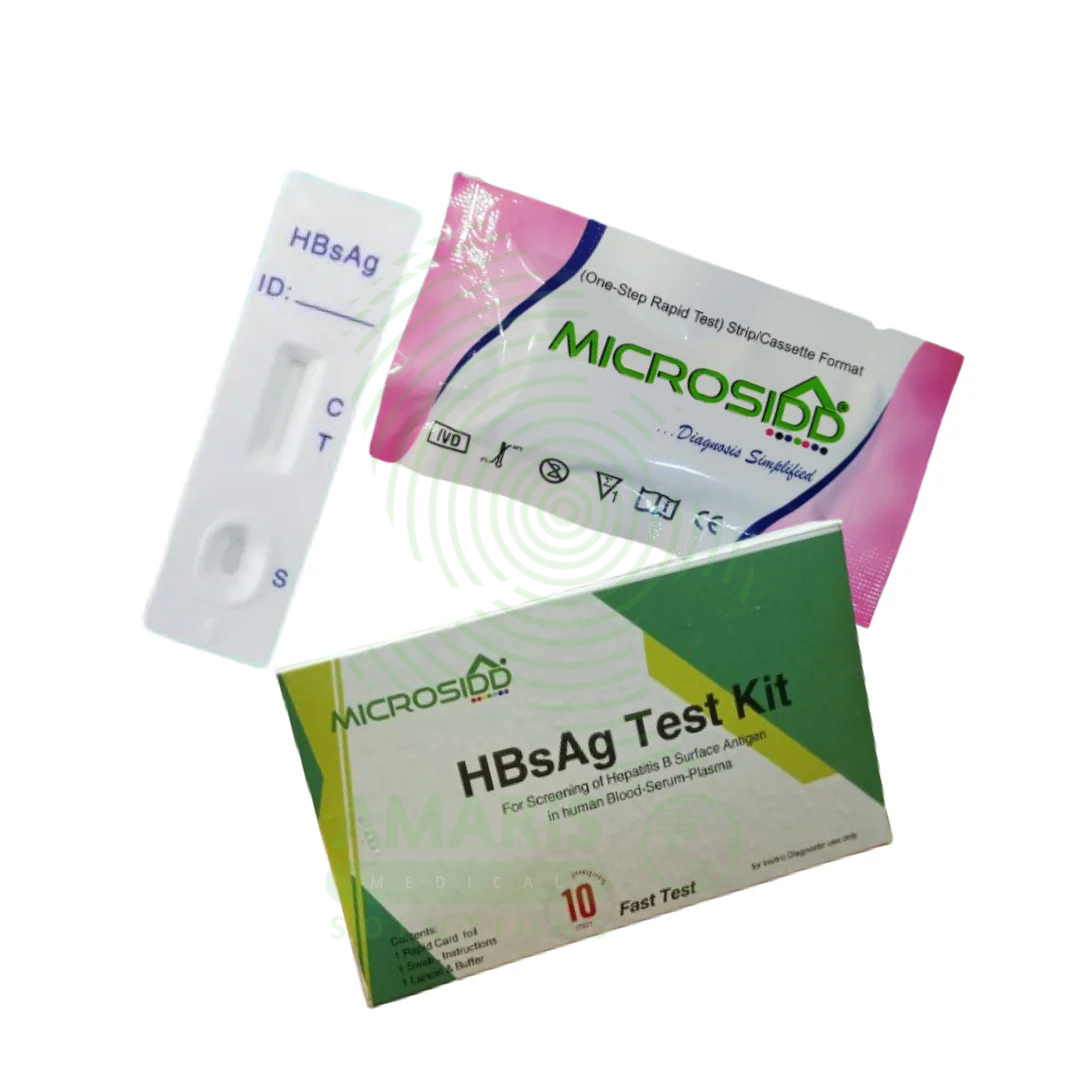
The Hepatitis B Test Kit is an in vitro diagnostic device for the detection of Hepatitis B virus markers in human blood samples, available in rapid, ELISA, and molecular formats. It detects specific antigens (HBsAg) and antibodies (anti-HBc, anti-HBs) to diagnose acute and chronic infection, monitor treatment response, screen blood donations, and determine immunity status. With high sensitivity (>98%) and specificity (>99%), it provides results in 15 minutes to 4 hours depending on format. Essential applications include prenatal screening to prevent mother-to-child transmission, evaluation of acute hepatitis, pre-immunosuppression screening, and occupational health testing. All patient samples must be handled with universal precautions, and testing must include appropriate quality controls.
Categories: CONSUMABLES AND DISPOSABLES, DIAGNOSTIC EQUIPMENT, Diagnostic Kits, Lab Consumables, LABORATORY EQUIPMENT AND SUPPLIES, Swabs, Applicators, and Sponges
Tags: Bloodborne Pathogen Testing, HBsAg Rapid Test, HBV Test, Hepatitis B Screening, Hepatitis B Test, Point Of Care Test, Viral Hepatitis Diagnosis
Description
Hepatitis B Test Kit
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Screening and Diagnosis of Hepatitis B Infection
-
Primary Use: Detects specific antigens and antibodies in blood to diagnose acute or chronic Hepatitis B virus infection, including HBsAg (surface antigen), anti-HBc (core antibody), and anti-HBs (surface antibody), enabling differentiation between acute, chronic, and resolved infection.
-
How it helps: For the hepatologist, infectious disease specialist, and primary care provider, the Hepatitis B test kit provides the serological roadmap essential for understanding a patient’s HBV status—revealing whether they have an active infection, are chronic carriers, have resolved past infection, or have developed protective immunity through vaccination. For the patient, these test results determine whether they need treatment, monitoring, or vaccination, and provide critical information about their risk of transmitting the virus to others.
2. Prenatal Screening to Prevent Mother-to-Child Transmission
-
Primary Use: Essential for screening pregnant women for Hepatitis B infection as part of routine prenatal care, allowing for intervention at birth to prevent vertical transmission to the newborn.
-
How it helps: For the obstetrician and pediatrician, prenatal HBV screening is a critical public health intervention—identifying infected mothers so that newborns can receive hepatitis B immune globulin and the first dose of vaccine within 12 hours of birth, reducing the risk of chronic infection from 90% to less than 5%. For the infant born to an infected mother, this testing and subsequent prophylaxis means they can be protected from a lifetime of chronic liver disease and cancer risk.
3. Monitoring Disease Progression and Treatment Response
-
Primary Use: Serial testing of HBV DNA viral load and liver enzymes guides treatment decisions, monitors response to antiviral therapy, and detects the development of drug resistance in patients undergoing treatment for chronic Hepatitis B.
-
How it helps: For the gastroenterologist and hepatologist managing patients on antiviral therapy, quantitative HBV testing provides the objective data needed to assess treatment efficacy—declining viral loads indicate response, rising levels may signal resistance or non-adherence. For the patient living with chronic Hepatitis B, regular monitoring means their treatment can be optimized, complications can be prevented, and they can have confidence that their disease is being managed effectively.
4. Screening High-Risk Populations and Blood Donations
-
Primary Use: Used to screen individuals in high-risk groups and to test all donated blood and blood products for Hepatitis B to prevent transmission through transfusion.
-
How it helps: For the blood bank director and public health official, universal screening of donated blood for HBV has nearly eliminated transfusion-transmitted Hepatitis B, protecting countless recipients from infection. For the individual receiving blood products during surgery, trauma, or medical treatment, this screening means they can receive life-saving transfusions without fear of acquiring a chronic viral infection.
5. Determination of Immunity and Vaccination Need
-
Primary Use: Quantitative anti-HBs testing determines whether an individual has protective immunity following vaccination or past infection, and identifies those who require vaccination or booster doses.
-
How it helps: For the occupational health physician and travel medicine specialist, anti-HBs testing confirms whether healthcare workers, travelers, and other at-risk individuals are protected against HBV—guiding decisions about vaccination, boosters, and work restrictions. For the healthcare worker at risk of occupational exposure, knowing their immunity status provides peace of mind and guides post-exposure prophylaxis decisions.
SECONDARY & SUPPORTIVE USES
1. Evaluation of Acute Hepatitis Presentations: When a patient presents with jaundice, elevated liver enzymes, or symptoms of acute hepatitis, HBV testing is essential for determining the cause and guiding management. For the patient with unexplained liver inflammation, rapid HBV testing provides answers and directs appropriate treatment.
2. Pre-Immunosuppression Screening: Before initiating immunosuppressive therapy for conditions like cancer, autoimmune disease, or organ transplantation, HBV screening identifies patients at risk of viral reactivation, allowing for prophylactic antiviral therapy. For the patient about to undergo chemotherapy or transplant, pre-treatment screening prevents the potentially fatal complication of HBV reactivation.
3. Contact Tracing and Family Screening: When an individual is diagnosed with Hepatitis B, testing of household contacts and sexual partners identifies others who may be infected and need treatment, or who are susceptible and need vaccination. For family members of an infected individual, this testing provides the information they need to protect themselves.
4. Pre-Employment and Occupational Health Screening: For healthcare workers, first responders, and others with occupational exposure risk, baseline HBV testing establishes immunity status and guides vaccination programs. For the employee entering a high-risk field, knowing their HBV status protects both themselves and their future patients.
5. Research and Epidemiological Surveillance: Used in clinical research and public health surveillance to track the prevalence, incidence, and epidemiology of Hepatitis B infection in populations. For the public health official, this data guides prevention and control strategies that protect entire communities.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: An in vitro diagnostic device for the qualitative or quantitative detection of Hepatitis B virus markers in human blood samples.
-
Common Names: Hepatitis B Test, HBV Test Kit, HBsAg Rapid Test, Hepatitis B Panel, Anti-HBs Test.
-
Test Types:
-
Rapid Diagnostic Tests: Point-of-care, lateral flow immunochromatographic assays providing results in 15-20 minutes.
-
ELISA Tests: Enzyme-linked immunosorbent assays for high-throughput laboratory screening with enhanced sensitivity and specificity.
-
Chemiluminescence Immunoassays: Automated, quantitative tests with high sensitivity for core laboratory use.
-
Nucleic Acid Tests: PCR-based tests for detecting HBV DNA, used for viral load monitoring and blood donor screening.
-
-
Sample Types: Whole blood, serum, or plasma depending on the specific test format.
-
Core Technology: Immunoassay (antigen-antibody reaction) or molecular amplification (for NAT).
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Sensitivity: Clinical sensitivity >98% for most FDA-approved tests, detecting HBsAg at concentrations as low as 0.1-0.5 IU/mL.
-
Specificity: Clinical specificity >99%, minimizing false positive results that would cause unnecessary anxiety and additional testing.
-
Time to Result: 15-20 minutes for rapid tests; 1-4 hours for ELISA; variable for automated platforms.
-
Storage Requirements: Typically 2-30°C; some components may require refrigeration at 2-8°C.
-
Shelf Life: 12-24 months depending on the specific test and storage conditions.
-
Controls: Built-in procedural controls to validate test performance and reagent integrity.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Rapid Test Format: Individually pouched test cassettes or strips with desiccant, including pipette for sample application and buffer vial.
-
ELISA Format: Microtiter plates pre-coated with antigens or antibodies, including wash buffers, conjugates, substrates, and stop solution.
-
Interpretation: Visual reading for rapid tests; spectrophotometric reading for ELISA and automated platforms.
-
Packaging: Available in various kit sizes from individual tests to bulk laboratory packs.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II or III medical device (depending on jurisdiction) with FDA 510(k) clearance or CE marking for diagnostic use.
-
Quality Management: Manufactured under ISO 13485 certified processes.
-
Intended Use: For professional use in laboratory or point-of-care settings.
-
Biohazard Precautions: All patient samples should be handled as potentially infectious. Universal precautions required.
-
Disposal: Used tests and samples must be disposed of as biohazardous waste.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage Conditions: Store in a cool, dry place at specified temperature range. Do not freeze unless indicated. Protect from direct sunlight and moisture.
-
Handling: Bring all reagents and samples to room temperature before testing. Do not use expired kits. Avoid cross-contamination of samples.
-
Disposal: Dispose of all used tests, samples, and pipettes in biohazard containers according to local regulations.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Screening, diagnosis, and monitoring of Hepatitis B virus infection in clinical, public health, and blood bank settings.
-
Workflow Integration: Suitable for point-of-care testing in clinics, emergency departments, and outreach settings, as well as high-volume automated testing in central laboratories.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Universal Precautions: Treat all patient samples as potentially infectious. Wear appropriate personal protective equipment (gloves, lab coat, eye protection).
-
Handling: Do not eat, drink, or smoke in the testing area. Wash hands thoroughly after handling samples and tests.
-
Disposal: Dispose of all contaminated materials in biohazard waste containers.
-
Quality Control: Run positive and negative controls as required to ensure test performance.
-
Interpretation: Read results within the specified time window. Delayed reading may yield inaccurate results.
2. FIRST AID MEASURES
-
Sample Splash to Skin: Wash thoroughly with soap and water. Follow institutional protocol for bloodborne pathogen exposure.
-
Sample Splash to Eyes: Flush eyes with copious amounts of clean water or saline for at least 15 minutes. Seek medical attention.
-
Needlestick Injury: If injury occurs with a contaminated lancet or device, wash the area thoroughly, apply antiseptic, and follow institutional post-exposure prophylaxis protocol.
3. FIRE FIGHTING MEASURES
-
Flammability: Reagents may contain alcohols or other flammable components. Keep away from open flames and sparks.
-
Extinguishing Media: Use alcohol-resistant foam, CO₂, or dry chemical extinguishers.
-
Spill Response: In case of large reagent spills, evacuate area, eliminate ignition sources, and follow institutional chemical spill protocol.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties