Patient Monitor
WhatsApp Order
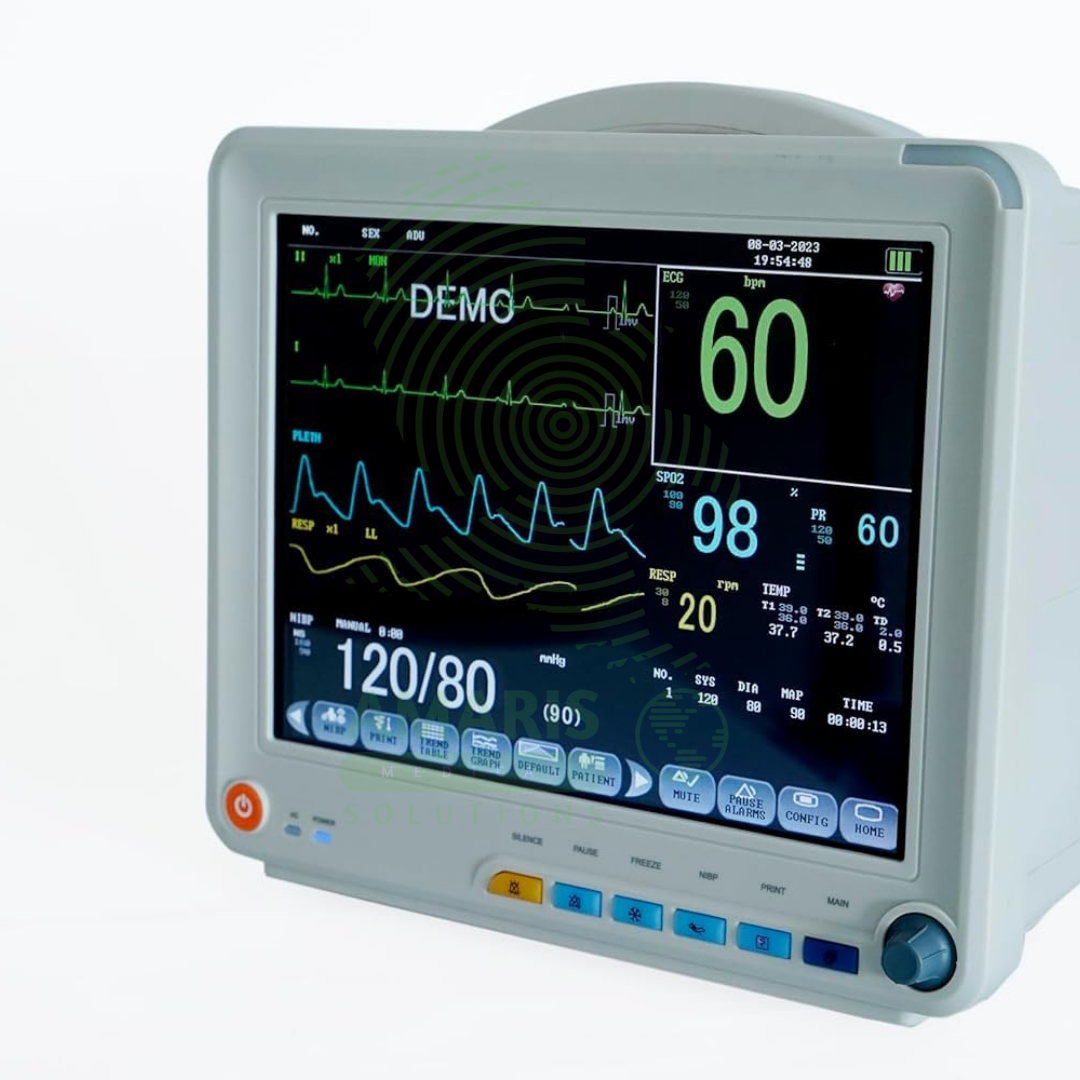
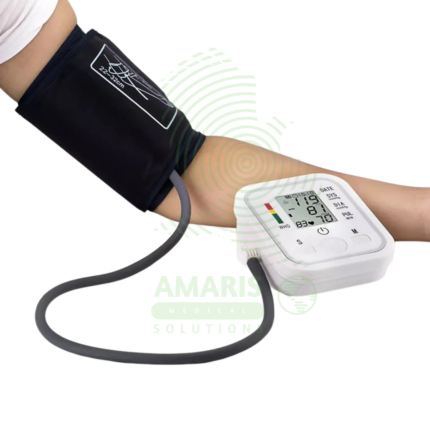
The Patient Monitor (6 Parameters BCCMS8000) is a versatile multi-parameter monitor designed for continuous surveillance of core vital signs in various clinical settings. It tracks six essential parameters—ECG, SpO2, Non-Invasive Blood Pressure (NIBP), Respiration, Temperature, and Pulse Rate—providing clinicians with real-time waveforms and numerical data on a clear color display. With its robust alarm system, battery backup for transport, and reliable performance, it is a fundamental tool for ensuring patient safety on general hospital wards, during procedures, and in emergency departments. Its design balances comprehensive monitoring capability with user-friendly operation.
Description
Patient Monitor
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Continuous Vital Signs Monitoring in General Ward Settings:
-
Primary Use: The primary use is for the non-invasive, continuous monitoring of core physiological parameters for stable but at-risk inpatients on medical-surgical floors, step-down units, and post-operative recovery areas.
-
How it helps: Keeps a constant watchful eye on patients who are stable enough to be out of the ICU but still need close observation, alerting nurses immediately if a patient’s heart rate, blood pressure, or oxygen levels begin to drift into dangerous territory.
2. Anesthesia & Procedural Sedation Monitoring:
-
Primary Use: Serves as a standard of care during surgical procedures under anesthesia and for moderate sedation during endoscopic or other bedside procedures to ensure patient stability.
-
How it helps: Provides the anesthesia team with real-time feedback on how a patient’s body is responding to surgery and anesthesia, allowing them to make split-second adjustments to keep the patient safe and stable throughout the procedure.
3. Emergency Department Triage & Observation:
-
Primary Use: Provides immediate and continuous assessment of patients in the ED, from initial triage through observation stays, allowing for rapid detection of clinical deterioration.
-
How it helps: Helps emergency physicians prioritize care by continuously tracking vital signs, ensuring that a patient whose condition is worsening is immediately identified and moved up for treatment.
4. Inter-Hospital & Intra-Hospital Patient Transport:
-
Primary Use: Its portable design makes it essential for safely monitoring critically ill or unstable patients during transfers between departments (e.g., to radiology) or between facilities.
-
How it helps: Extends the safety net of the ICU or ED during transport, ensuring that a patient’s condition is monitored every second they are away from their unit, with alarms that alert the transport team to any problems.
5. High-Dependency Patient Monitoring:
-
Primary Use: Used for patients who require closer observation than standard ward care but do not yet need intensive care, such as those on specific medication drips, with worsening chronic conditions, or post-complex procedures.
-
How it helps: Bridges the gap between general ward care and intensive care, providing a higher level of monitoring for patients who need it without the cost and resource intensity of an ICU bed.
SECONDARY & SUPPORTIVE USES
1. Cardiac Stress Testing Support: Often used in conjunction with treadmill or pharmacological stress tests to monitor ECG and hemodynamic responses in real-time, helping doctors assess how the heart responds to exertion.
2. Respiratory Therapy & Oxygen Titration: Assists in titrating oxygen therapy by providing continuous SpO2 readings and respiratory rate monitoring for patients with COPD, pneumonia, or other respiratory conditions, ensuring they receive exactly the right amount of oxygen.
3. Long-term Care & Sub-Acute Facility Monitoring: Provides a level of monitoring in skilled nursing facilities (SNFs) or rehabilitation centers for patients with significant cardiac or respiratory comorbidities, extending hospital-level monitoring to long-term care settings.
4. Clinical Training & Education: Used as a teaching tool for nursing and medical students to understand normal and abnormal vital sign trends and alarm management, training the next generation of healthcare providers.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Name: Patient Monitor
-
Type: Multi-Parameter Patient Monitor (MPM)
-
Monitoring Parameters: ECG, SpO2 (Pulse Oximetry), NIBP (Non-Invasive Blood Pressure), Respiration, Temperature, and likely Pulse Rate (derived from ECG/SpO2).
-
Display: Integrated color TFT/LCD screen (typically 12.1-inch or similar) for real-time waveform and numerical display.
-
Form Factor: Designed as a portable unit with a carrying handle or as a compact bedside monitor, often with a modular docking system.
2. TECHNICAL & PERFORMANCE PROPERTIES
-
ECG Monitoring: Typically 3 or 5-lead configuration. Features include heart rate calculation, arrhythmia detection (PVCs, tachycardia, bradycardia), ST-segment analysis, and multiple selectable filter modes (monitor, diagnostic, filter).
-
SpO2 Monitoring: Measures arterial oxygen saturation and pulse rate via finger probe. Includes plethysmograph waveform and adjustable alarm limits for low saturation.
-
NIBP Monitoring: Automated oscillometric measurement of systolic, diastolic, and mean arterial pressure. Features include manual/auto cycling, STAT mode, and adjustable cuff sizes.
-
Respiration Monitoring: Typically measured via impedance pneumography through the ECG electrodes (thoracic impedance changes).
-
Temperature Monitoring: Via a single-use or reusable skin or rectal probe.
-
Alarm System: Comprehensive visual and audible alarms for each parameter with priority levels (e.g., high, medium, low). Includes alarm pause and sound level adjustment.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Power Supply: Dual power source - operates on AC mains power and a built-in rechargeable battery for uninterrupted monitoring during transport or power loss.
-
Battery Life: Typically provides 3 to 5 hours of continuous operation on a full charge.
-
Connectivity: Standard ports include USB for data export, Ethernet or optional WiFi for network connectivity to a Central Station Monitoring System.
-
User Interface: Combination of front-panel buttons, knobs, and/or a touchscreen for intuitive navigation and parameter setup.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Approvals: Must carry CE Marking (for EU market) and/or FDA 510(k) Clearance (for US market). Compliance with medical device directives (MDD/MDR) is essential.
-
Safety Standards: Complies with IEC 60601-1 (General Safety), IEC 60601-1-2 (Electromagnetic Compatibility), and IEC 60601-2-27 (Particular requirements for ECG monitoring equipment).
-
Protection Class: Type CF applied part for defibrillation protection on ECG leads.
5. STORAGE & HANDLING ATTRIBUTES
-
Environmental Conditions: Designed for hospital use. Operating temperature typically 10°C to 40°C, humidity 30% to 75% non-condensing. Storage temperature range is wider.
-
Cleaning & Disinfection: The casing should be cleaned regularly with a soft cloth dampened with hospital-grade disinfectant (e.g., diluted bleach or alcohol-based solutions compatible with plastics). Avoid liquid ingress into ports or vents.
-
Calibration: Requires periodic performance verification and calibration as per the manufacturer's service schedule and local regulatory requirements.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Continuous physiological surveillance to detect and alert caregivers to early signs of patient deterioration across various hospital departments.
-
Supportive Application: Provides documented vital sign trends for patient records, supports clinical decision-making, and is integral to patient safety protocols.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Personal Protective Equipment (PPE): Standard precautions apply. Gloves should be worn when applying ECG electrodes or if there is risk of contact with bodily fluids.
-
Patient Safety: Ensure correct electrode placement for accurate ECG and respiration. Use appropriate-sized blood pressure and SpO2 cuffs/probes to avoid injury or inaccurate readings. Inspect skin under probes periodically.
-
Device & Electrical Safety: Use only manufacturer-approved accessories (cuffs, probes, cables). Do not use if the housing is cracked or damaged. Ensure proper grounding and do not use in the presence of flammable anesthetics.
2. FIRST AID MEASURES
-
General: Patient care takes priority. In an emergency, disconnect non-essential monitoring if it impedes care.
-
Skin Irritation: Remove the offending electrode or probe. Clean skin with soap and water. Seek medical advice for persistent reactions.
-
Device Malfunction: If the monitor malfunctions (e.g., overheats, smokes), disconnect it from the patient and power source immediately. Replace with another monitor.
3. FIRE FIGHTING MEASURES
-
Flammability: Contains plastics, electronic components, and a lithium battery, all of which are combustible.
-
Extinguishing Media: For electrical fires, use CO₂ or dry powder extinguishers. Do not use water jets directly on the device.
-
Hazardous Combustion Products: Burning electronics can release toxic fumes (e.g., carbon monoxide, hydrogen fluoride from batteries).
-
Firefighter Instructions: Wear full protective gear and SCBA. Remove the device from the vicinity if safe to do so.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties