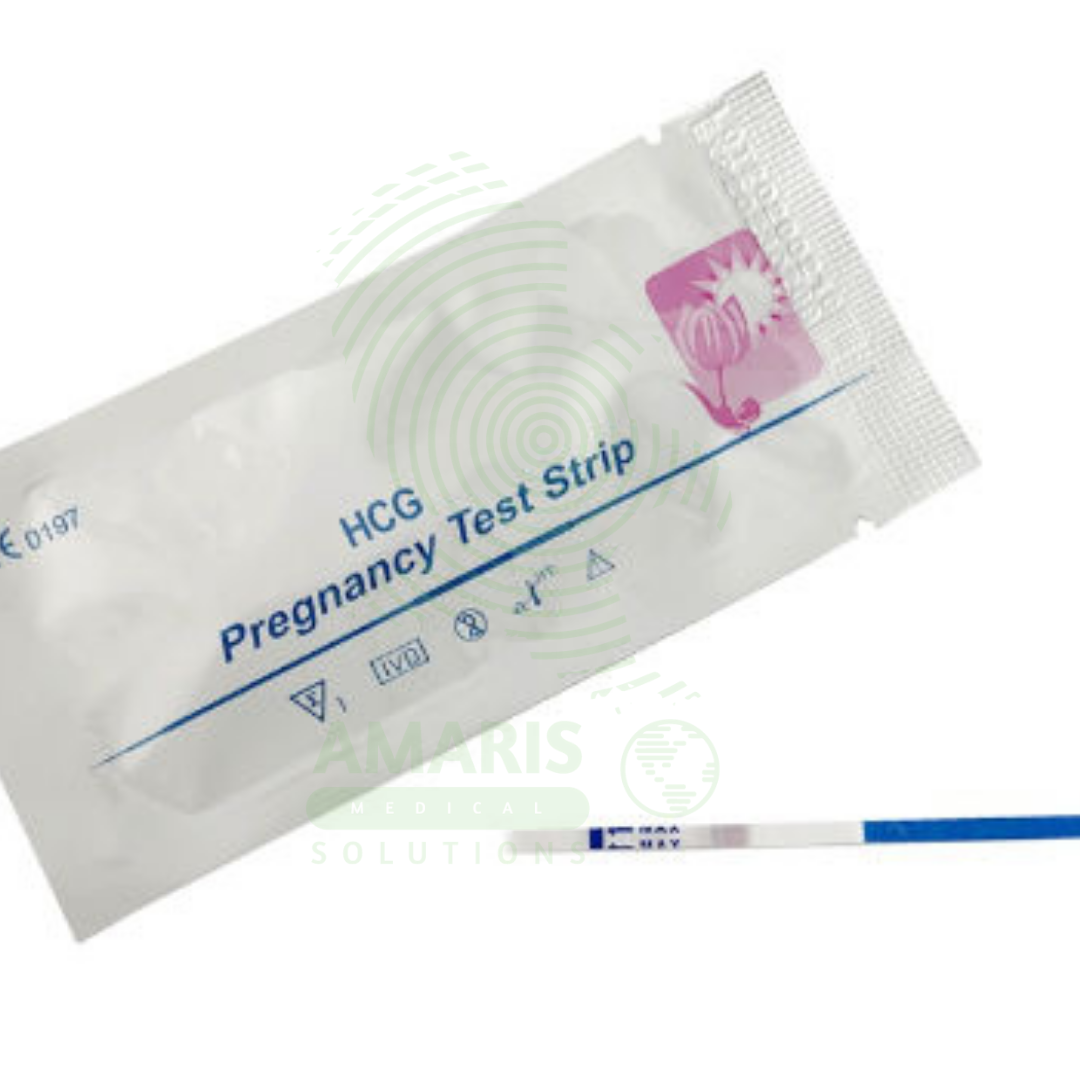
Pregnancy Test Strips
WhatsApp Order
Pregnancy Test Strips are simple, dip-style lateral flow devices designed for the rapid, qualitative detection of human chorionic gonadotropin (hCG) in urine to indicate pregnancy. As the most fundamental format of home pregnancy tests, they offer high sensitivity (often from 10 mIU/mL), affordability, and a quick result within 3-5 minutes. Their ease of use makes them a widely accessible first step in pregnancy detection for personal use and clinical screening. A visible test line, even if faint, is a positive result, though confirmation with a healthcare provider is always recommended.
Description
Pregnancy Test Strips
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Early Detection of Pregnancy:
-
Primary Use: The primary and most common use is for the early, qualitative detection of human chorionic gonadotropin (hCG) in urine to confirm pregnancy. This is used by individuals at home for personal knowledge and by healthcare professionals in clinics for initial assessment.
-
How it helps: Provides millions of people with a simple, private way to answer a life-changing question at home, giving them information they need to make important decisions about their health and future.
2. Clinical Confirmation of Pregnancy:
-
Primary Use: Used in physician offices, family planning clinics, and emergency departments to quickly confirm pregnancy as part of routine patient intake, pre-procedural workups, or when evaluating abdominal pain or other pregnancy-related symptoms.
-
How it helps: Gives healthcare providers immediate, essential information that guides every subsequent decision—from which medications are safe to prescribe to whether certain imaging studies should be performed.
3. Aiding in Reproductive Health Management:
-
Primary Use: Provides immediate information that is critical for making timely decisions regarding prenatal care initiation, discussing options with a healthcare provider, or managing conditions that may be affected by pregnancy.
-
How it helps: Empowers individuals to take control of their reproductive health by providing early confirmation that allows them to seek prenatal care, make informed choices, and manage their health appropriately from the earliest possible moment.
SECONDARY & SUPPORTIVE USES
1. Fertility Treatment Monitoring: Used in conjunction with fertility treatments (like IVF or ovulation induction) to detect pregnancy following embryo transfer or after a missed period, providing anxious couples with news they have been hoping for.
2. Rule-Out in Medical Situations: Used to rapidly rule out pregnancy as a cause of symptoms (e.g., amenorrhea, nausea) or before initiating medical treatments, procedures, or imaging studies that could be harmful to a fetus (e.g., certain medications, X-rays), protecting both patient and potential pregnancy.
3. Educational and Counseling Tool: Used in sexual health education and counseling settings to teach about reproductive health and pregnancy testing, helping individuals understand their options and make informed decisions.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: Rapid, qualitative, immunochromatographic urine test.
-
Common Names: Pregnancy Test Strip, Urine hCG Test, Home Pregnancy Test (HPT).
-
Target Analyte: Detects the beta-subunit of human Chorionic Gonadotropin (hCG), a hormone produced by the placenta shortly after embryo implantation.
-
Format: Typically a thin, rectangular strip of porous nitrocellulose membrane with defined test and control zones. Often sold in a simple foil pouch without a plastic casing (as opposed to midstream or cassette tests).
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Principle: Lateral Flow Immunoassay. The strip is impregnated with colored particles (e.g., colloidal gold) conjugated to anti-hCG antibodies. When the strip is dipped in urine, the urine migrates upward. If hCG is present, it forms a complex with the conjugate, which is then captured by a second line of immobilized anti-hCG antibodies in the test zone (T), forming a visible colored line.
-
Sensitivity: Critical performance characteristic. Measured in mIU/mL (milli-International Units per milliliter). Common sensitivities for early detection strips range from 10 mIU/mL to 25 mIU/mL. A lower number indicates higher sensitivity and the ability to detect pregnancy earlier (often 3-4 days before the expected period).
-
Procedure: Collect urine in a clean cup. Dip the test strip vertically into the urine to the marked line for the specified time (e.g., 5-10 seconds). Remove, lay flat, and read results after the specified wait time (e.g., 3-5 minutes).
-
Interpretation:
-
Positive: Two distinct colored lines appear—one in the Control region (C) and one in the Test region (T). Any visible line in the T region, even if faint, is considered positive.
-
Negative: Only one colored line appears in the Control region (C). No line appears in the Test region.
-
Invalid: No line appears in the Control region (C), regardless of the T line. The test is faulty and must be repeated with a new strip.
-
-
Time to Result: Results are typically readable in 3 to 5 minutes. Results should not be read after 10 minutes, as an evaporation line may appear.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Packaging: Individually sealed in a foil pouch with a desiccant to protect from moisture.
-
Simplicity and Cost: The most basic and economical format for hCG testing, requiring only the strip and a urine sample.
-
No Instrumentation Required: Visual readout.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Regulated as a Class II medical device in most markets. Requires FDA 510(k) clearance in the US and CE Marking in the EU for both professional and over-the-counter (OTC) use.
-
Accuracy Claims: Must meet stringent performance standards (e.g., >99% accuracy from the day of the expected period, as commonly advertised).
-
Quality Standards: Manufactured under ISO 13485.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in a cool, dry place at room temperature. Do not freeze. Protect from direct sunlight and moisture. Use before the expiration date printed on the pouch.
-
Handling: Remove the strip from the pouch just before use. Handle by the end opposite the absorbent tip to avoid contamination. Do not reuse.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: A highly accessible, rapid screening tool for pregnancy detection. It is a presumptive test.
-
Confirmatory Testing: A positive result should be followed by consultation with a healthcare provider, who may confirm with a clinical urine test, a quantitative serum hCG blood test, and ultimately, ultrasound.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Timing of Test: For highest accuracy, test with first-morning urine, which has the highest concentration of hCG. Testing too early after implantation can result in a false negative.
-
Read Instructions: Users must follow the instructions precisely regarding dipping time and reading time.
-
Interpretation of Faint Lines: A very faint line in the test region is usually a true positive indicating early pregnancy with low hCG levels. The test should be repeated in 2-3 days when hCG levels have doubled, resulting in a darker line.
-
Medication Interference: Certain medications containing hCG (e.g., some fertility drugs) can cause false positives. Most other medications do not interfere.
2. FIRST AID MEASURES
-
Ingestion: If the test strip is ingested, seek medical advice.
-
Chemical Exposure: The strip contains biologically inert materials. If particles cause eye irritation, rinse with water.
3. FIRE FIGHTING MEASURES
-
Flammability: The paper and plastic components are combustible.
-
Extinguishing Media: Use water, foam, or CO₂ as appropriate for the surrounding fire.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties