Water Purification Machine
WhatsApp Order
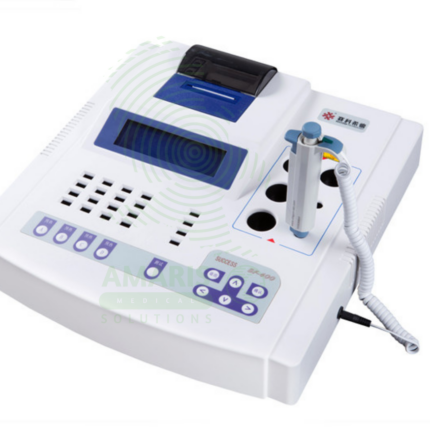
A Water Purification Machine produces high-purity water for clinical laboratory testing, hemodialysis, sterile processing, and pharmacy compounding. Using technologies such as reverse osmosis, deionization, and ultrafiltration, it removes ions, organic compounds, bacteria, and endotoxins to meet the stringent purity standards required for accurate diagnostic testing and safe patient care. Essential for clinical laboratories, dialysis units, and sterile processing departments, it provides the purified water critical for reliable test results and patient safety.
Description
Water Purification Machine
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Production of Purified Water for Clinical Laboratory Testing
-
Primary Use: Produces high-purity water for clinical chemistry, immunoassay, molecular diagnostics, and microbiology laboratories. The purification system removes ions, organic compounds, bacteria, and endotoxins, providing water that meets the stringent purity requirements for accurate diagnostic testing.
-
How it helps: For the laboratory manager and clinical scientist, the water purification machine ensures that all water used in testing meets required purity standards—eliminating variables that could compromise test accuracy. For the patient, purified water contributes to reliable test results that guide diagnosis, treatment, and monitoring.
2. Water for Reagent Preparation and Calibration
-
Primary Use: Provides purified water for preparing reagents, calibrators, controls, and standards used in diagnostic testing. Consistent water quality ensures that reagent performance is reproducible and that calibration curves are accurate.
-
How it helps: For the laboratory technician, purified water provides the consistent matrix needed for accurate calibration and quality control—ensuring that patient results are reliable and comparable across testing platforms. For the patient, accurate test results depend on the quality of water used in reagent preparation.
3. Rinsing of Laboratory Glassware and Analytical Equipment
-
Primary Use: Purified water is used for final rinsing of laboratory glassware, cuvettes, and analytical equipment to prevent mineral deposits and contamination that could interfere with sensitive measurements.
-
How it helps: For the laboratory staff, purified water rinsing ensures that glassware and equipment are free of residues that could affect test results or cause instrument malfunction. For the patient, clean, residue-free equipment contributes to accurate diagnosis.
4. Water for Hemodialysis Treatment
-
Primary Use: Produces purified water for hemodialysis machines, meeting the strict purity standards required for dialysis water to prevent patient exposure to contaminants, bacteria, and endotoxins. Water purification is critical for patient safety in dialysis.
-
How it helps: For the dialysis unit and biomedical engineering team, water purification systems ensure that dialysis water meets safety standards—protecting patients from pyrogenic reactions, bacteremia, and long-term complications. For the patient receiving dialysis, purified water is essential for safe and effective treatment.
5. Water for Sterile Processing and Instrument Cleaning
-
Primary Use: Provides purified water for final rinsing of surgical instruments, endoscopes, and medical devices after cleaning, preventing mineral deposits that can interfere with sterilization or damage delicate instruments.
-
How it helps: For the sterile processing department, purified water rinsing ensures that instruments are free of residues that could compromise sterilization or cause instrument damage. For the patient, properly processed instruments reduce the risk of healthcare-associated infections.
SECONDARY & SUPPORTIVE USES
1. Water for Pharmacy Compounding: Purified water for compounding sterile and non-sterile preparations.
2. Water for Clinical Chemistry Analyzers: Feed water for automated clinical chemistry and immunoassay analyzers.
3. Water for Histology and Cytology: Purified water for tissue processing, staining, and mounting procedures.
4. Water for Dental Unit Waterlines: Purified water for dental procedures to prevent biofilm formation.
5. Water for Laboratory Animal Facilities: Purified water for animal watering systems.
6. Water for Research and Development: High-purity water for pharmaceutical and biotechnology research.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: A water purification system that produces high-purity water for clinical, laboratory, and healthcare applications.
-
Designation: Water Purification Machine, Water Purification System, Reverse Osmosis System, Deionization System, Clinical Water System.
-
Purification Technologies:
-
Reverse Osmosis: Removes dissolved salts, organics, and microorganisms.
-
Deionization: Removes ions using ion exchange resins.
-
Ultrafiltration: Removes endotoxins and bacteria.
-
UV Disinfection: Inactivates microorganisms.
-
Distillation: Boiling and condensation for high purity.
-
-
Key Components:
-
Pre-Filters: Sediment and carbon filtration.
-
Reverse Osmosis Membrane: Primary purification stage.
-
Deionization Cartridges: Ion exchange for high purity.
-
UV Lamp: Ultraviolet disinfection.
-
Storage Tank: Purified water reservoir.
-
Distribution Loop: Recirculating system to maintain purity.
-
Conductivity Monitor: Continuous water quality monitoring.
-
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Water Quality: 0.1-18.2 MΩ·cm resistivity depending on system.
-
TOC (Total Organic Carbon): <1-10 ppb depending on system.
-
Bacteria: <1 CFU/mL for clinical systems.
-
Endotoxins: <0.03 EU/mL for dialysis systems.
-
Production Rate: 10-1000 liters per hour depending on model.
-
Storage Capacity: 50-500 liters depending on configuration.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Configuration: Benchtop, freestanding, or wall-mounted.
-
Installation: Professional installation required; water and electrical connections.
-
Controls: Digital interface with water quality monitoring.
-
Portability: Stationary installation.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class I or Class II medical device depending on application.
-
Water Quality Standards: Compliant with CLSI, AAMI, and USP standards.
-
Electrical Safety: Compliant with electrical safety standards.
-
Materials: FDA-compliant materials for water contact.
5. STORAGE & HANDLING ATTRIBUTES
-
Installation: Professional installation required; proper water and electrical connections.
-
Maintenance: Regular filter and membrane replacement; sanitization cycles.
-
Water Quality Monitoring: Continuous conductivity monitoring; periodic testing.
-
Documentation: Maintenance logs and water quality records.
6.LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Production of purified water for clinical laboratory testing, hemodialysis, and sterile processing.
-
Clinical Role: Essential for accurate diagnostic testing, safe dialysis, and effective instrument reprocessing.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Water Quality Monitoring: Monitor conductivity and bacteria levels regularly.
-
Filter Replacement: Replace filters and membranes per manufacturer schedule.
-
Sanitization: Perform regular sanitization to prevent biofilm formation.
-
Storage: Store purified water in appropriate containers; use within recommended timeframe.
-
Backflow Prevention: Ensure backflow prevention devices are installed.
2. FIRST AID MEASURES
-
Chemical Exposure: If chemicals (cleaners, sanitizers) contact skin or eyes, rinse with water; seek medical attention if irritation persists.
3. FIRE FIGHTING MEASURES
-
Flammability: Electrical components may pose fire risk.
-
Extinguishing Media: For electrical fire, use CO₂ or dry chemical extinguisher.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties