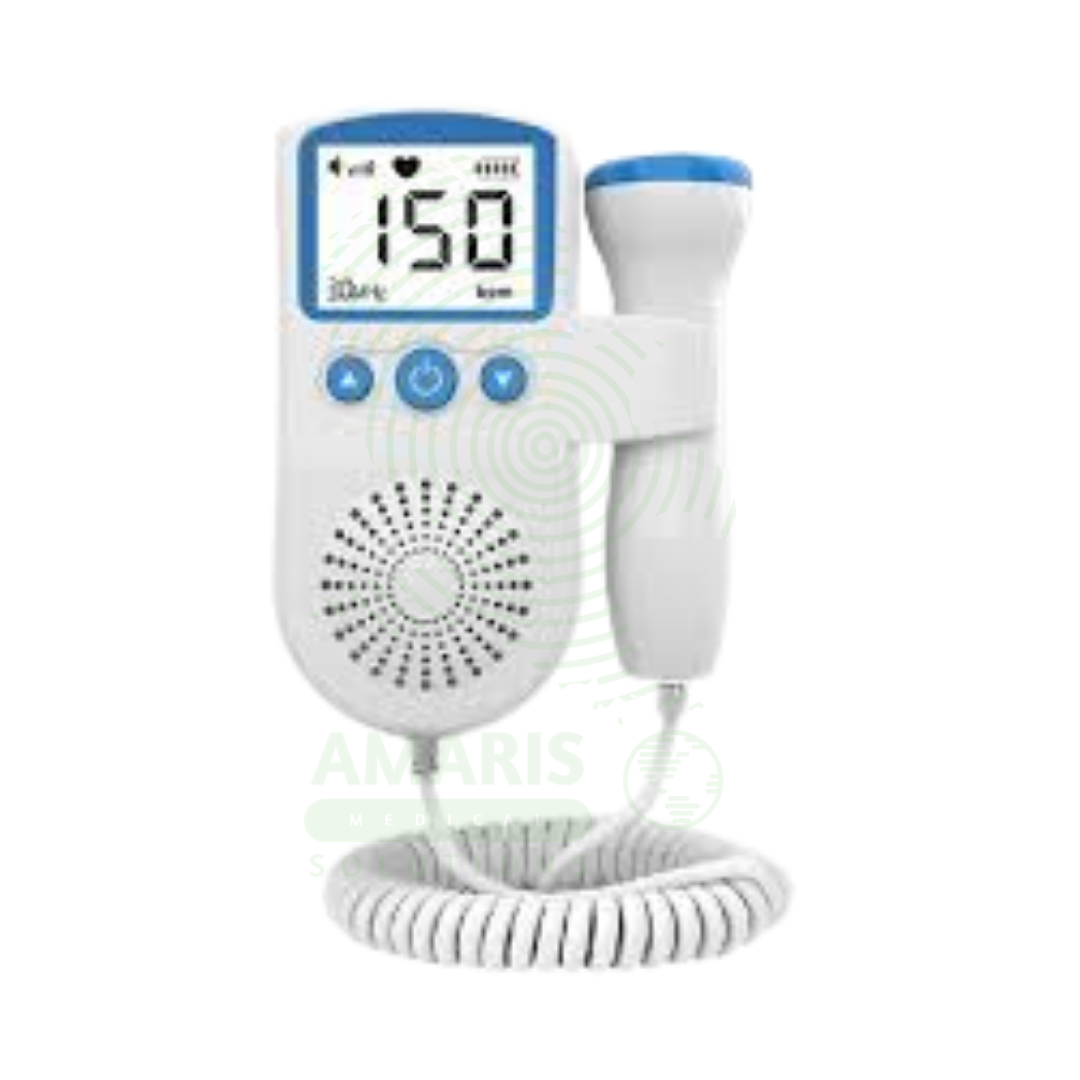
Fetoheart Sonic Aid Doppler (clinic/hospital use)
WhatsApp Order
A Fetoheart Sonic Aid Doppler (Fetal Doppler) is a Class II medical ultrasound device (2-3 MHz probe) for real-time detection and audible/visual display of fetal heart rate in clinical obstetric settings from 10-12 weeks gestation. Features include handheld or tabletop console, waterproof probe, digital heart rate display (30-240 bpm), amplified speaker, headphone jack, and rechargeable battery or AC power operation. Continuous wave or pulsed wave Doppler technology with auto-correlation signal processing rejects maternal artifact. Primary clinical applications include fetal heart rate detection and monitoring during routine prenatal visits, confirmation of fetal viability in first trimester, intermittent fetal monitoring during labor, non-stress test administration, multiple gestation assessment, fetal presentation determination, and post-procedure confirmation (amniocentesis, CVS, ECV). Critical safety considerations include following ALARA principle (minimum necessary exposure), appropriate ultrasound gel use, probe pressure avoidance, infection control (probe cleaning between patients), differentiation of fetal from maternal signals, and interpretation by qualified providers. Essential equipment in outpatient obstetric clinics, hospital labor and delivery units, and prenatal care settings.
Categories: DIAGNOSTIC EQUIPMENT, Diagnostic Kits, Fetal Dopplers and Monitors, Multi-Parameter Monitors, OBSTETRICS, GYNECOLOGY AND NEONATAL, PATIENT MONITORING AND LIFE SUPPORT
Tags: Dop tone, Fetal Doppler, Fetal Heart Detector, Fetal Monitor, Labor And Delivery, Obstetric Ultrasound, Prenatal Care
Description
Fetoheart Sonic Aid Doppler (clinic/hospital use)
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Fetal Heart Rate Detection and Monitoring:
-
Primary Use: Provides real-time audible and visual display of fetal heart rate using Doppler ultrasound technology, allowing clinicians to assess fetal cardiac activity, rate, and rhythm during routine prenatal visits from as early as 10-12 weeks gestation.
-
How it helps: Gives obstetricians and midwives an immediate window into the baby’s well-being at every prenatal visit, providing objective evidence that the pregnancy is progressing normally and alerting them to any concerns that need further investigation.
2. Confirmation of Fetal Viability:
-
Primary Use: Rapid, non-invasive confirmation of fetal cardiac activity in first-trimester pregnancies, particularly valuable for patients with history of miscarriage, bleeding, or cramping where viability assessment is clinically indicated.
-
How it helps: Provides anxious parents and their doctors with immediate answers during moments of uncertainty, offering reassurance that the pregnancy is viable when there are concerns about miscarriage.
3. Intermittent Fetal Monitoring During Labor:
-
Primary Use: Used in hospital labor and delivery units for periodic assessment of fetal heart rate between contractions in low-risk pregnancies, providing audible feedback of fetal response to labor.
-
How it helps: Allows laboring mothers to maintain mobility and a less restrictive birth experience while still ensuring their baby is regularly checked for any signs of distress.
4. Non-Stress Test (NST) Administration:
-
Primary Use: Integrated with fetal monitors for non-stress testing in outpatient obstetric clinics, assessing fetal heart rate accelerations in response to fetal movement as an indicator of fetal well-being.
-
How it helps: Provides reassurance that a baby in a high-risk pregnancy is healthy and receiving enough oxygen, helping doctors make informed decisions about delivery timing.
5. Multiple Gestation Assessment:
-
Primary Use: Allows identification and monitoring of individual fetal heart rates in twin and higher-order multiple pregnancies, with experienced clinicians able to distinguish between fetuses.
-
How it helps: Helps clinicians caring for mothers expecting multiples ensure each baby is developing normally, providing individual attention to every fetus in the womb.
6. Fetal Presentation and Position Determination:
-
Primary Use: The point of maximal intensity of fetal heart sounds helps experienced practitioners determine fetal lie and presentation, particularly in later gestation.
-
How it helps: Gives clinicians valuable information about how the baby is positioned before delivery, helping them plan for a safe birth and anticipate any potential complications.
7. Post-Procedure Confirmation:
-
Primary Use: Used after amniocentesis, chorionic villus sampling, or external cephalic version to confirm continued fetal well-being and cardiac activity.
-
How it helps: Provides immediate reassurance after invasive procedures that the baby is tolerating the intervention well and that fetal heart activity remains normal.
SECONDARY & SUPPORTIVE USES
1. Patient Reassurance: Provides audible fetal heart sounds to expectant parents during prenatal visits, enhancing bonding and reducing anxiety by letting them hear their baby’s heartbeat.
2. Teaching Tool: Used in obstetric education to teach students and residents fetal heart rate auscultation and interpretation, training the next generation of healthcare providers.
3. Triage Assessment: Rapid assessment of fetal heart rate in patients presenting to labor and delivery with decreased fetal movement or other concerns, helping prioritize care.
4. Preoperative Verification: Confirms fetal heart rate prior to cerclage placement or other obstetric procedures, ensuring the baby is stable before interventions.
5. Research Applications: Used in studies requiring fetal heart rate data collection, contributing to scientific understanding of fetal development and pregnancy complications.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Device Type: Handheld or tabletop Doppler ultrasound device for fetal heart rate detection.
-
Common Names: Fetal Doppler, Fetal Heart Detector, Fetal Monitor, Doptone, Fetal Sonic Aid.
-
Components:
-
Main Unit: Handheld or tabletop console with controls and display.
-
Probe/Transducer: 2-3 MHz waterproof probe with integrated ultrasound crystal.
-
Display: LCD showing fetal heart rate (numeric) and sometimes signal strength.
-
Speaker: Built-in speaker for audible heart sounds; headphone jack available.
-
Power Source: Rechargeable battery, AC power, or replaceable batteries.
-
-
Probe Frequencies: 2 MHz (deeper penetration, later gestation, obese patients), 3 MHz (better resolution, earlier gestation, thin patients).
-
Display Range: 30-240 bpm typical.
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Ultrasound Technology: Continuous wave or pulsed wave Doppler detecting motion of fetal heart structures.
-
Output Display: Digital numeric display of fetal heart rate (averaged over 3-6 beats).
-
Audio Output: Amplified heart sounds through speaker; adjustable volume.
-
Auto-Correlation: Signal processing to reject maternal vessel noise and artifact.
-
Depth Penetration: Up to 15-20 cm depending on frequency and tissue characteristics.
-
Detection Threshold: Capable of detecting fetal heart activity from 10-12 weeks gestation; earlier in thin patients, later in obese or with retroverted uterus.
-
Battery Life: 2-8 hours continuous use depending on model.
-
Wall-Plug Operation: Continuous use capability for labor and delivery settings.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Handheld Unit Dimensions: 15-20 cm × 5-8 cm × 3-5 cm (6-8 × 2-3 × 1-2 inches).
-
Weight: 200-500 grams (7-18 ounces) depending on battery configuration.
-
Probe: 10-15 cm length, 2-3 cm diameter; with 1.5-2 meter cable.
-
Display: 2-4 cm high LCD with large numerals.
-
Controls: On/off, volume, probe selection (if multi-probe), freeze (select models).
-
Speaker: 1-2 watts; clear audio in quiet clinical environment.
-
Headphone Jack: 3.5 mm for private listening or noisy environments.
-
Probe Storage: Built-in probe holder or separate storage compartment.
-
Carrying Case: Padded case for transport; wall-mountable bracket available.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device requiring FDA 510(k) clearance.
-
Intended Use: For detection of fetal heart rate during pregnancy and labor.
-
Ultrasound Safety: Output within FDA limits for diagnostic fetal use; mechanical index (MI) < 1.9, thermal index (TI) < 1.0.
-
Electrical Safety: Compliant with IEC 60601-1 for medical electrical equipment; Type BF applied part.
-
ALARA Principle: Device operates at lowest feasible output to obtain adequate signal.
-
Probe Disinfection: Waterproof probes allow cleaning with appropriate low-level disinfectant; probe covers available for specific indications.
-
Sterility: Probes non-sterile; not for intrauterine use.
-
EMC Compliance: IEC 60601-1-2 electromagnetic compatibility; immune to typical clinical interference.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in clean, dry environment in carrying case or on wall mount; protect probes from damage.
-
Battery Maintenance: Recharge after each use; maintain spare charged battery for continuous use settings.
-
Probe Care: Do not drop probes; inspect cable for damage; clean after each use with approved disinfectant wipe; do not immerse probe in liquids unless specified waterproof.
-
Cleaning: Wipe unit and probe with EPA-registered hospital disinfectant between patients.
-
Calibration: Annual calibration verification recommended per facility biomedical engineering protocol.
-
Inspection: Before each use, check probe cable, connector, and housing; verify audio output and display function.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Real-time fetal heart rate detection and monitoring in outpatient obstetric clinics, hospital labor and delivery units, and prenatal care settings.
-
Gestational Age Considerations:
-
10-12 weeks: Earliest detection in ideal conditions.
-
12-14 weeks: Reliable detection in most patients with appropriate technique.
-
14-16 weeks: Consistent detection in nearly all patients.
-
16+ weeks: Routine use for all prenatal visits.
-
-
Technique:
-
Apply ultrasound gel to maternal abdomen.
-
Position probe suprapubically, angled toward pelvis.
-
Slowly sweep until fetal heart tones identified.
-
Adjust angle for optimal signal.
-
Count rate (displayed) or auscultate for 60 seconds.
-
Limitations: May be difficult in early gestation, maternal obesity, retroverted uterus, or with anterior placenta; requires ultrasound gel for acoustic coupling.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Ultrasound Safety: Follow ALARA principle (As Low As Reasonably Achievable); use minimum power and duration necessary. No known adverse effects at diagnostic output levels for fetal use.
-
Gel Use: Always use appropriate ultrasound gel; never use with dry probe.
-
Probe Pressure: Avoid excessive pressure on maternal abdomen, especially in early pregnancy.
-
Infection Control: Clean probe between each patient; use disposable probe covers if probe will contact mucous membranes or if patient has known infection.
-
Interpretation: Fetal heart rate must be interpreted by qualified healthcare providers; rates <110 or >160 bpm warrant further evaluation.
-
Maternal Signal Differentiation: Distinguish fetal heart rate (typically 110-160 bpm) from maternal vessel signal (rate equals maternal pulse).
-
Doppler Not for Home Use: These clinical-grade devices intended for trained healthcare providers; not for unsupervised home use.
-
EMI Caution: May be affected by strong electromagnetic fields; avoid use near MRI.
2. FIRST AID MEASURES
-
Inability to Detect Fetal Heart: Reposition patient; try different probe angle; use different frequency probe if available; if persistent absence after 12 weeks with previous detection, arrange urgent ultrasound.
-
Gel in Eyes: Flush with copious water; seek medical attention if irritation persists.
-
Equipment Malfunction: Discontinue use; return for service; document malfunction.
-
Maternal Discomfort: Reposition; reduce probe pressure.
3. FIRE FIGHTING MEASURES
-
Flammability: Plastic casing and components are combustible.
-
Extinguishing Media: For electrical fire, use CO₂ or dry chemical (Class C) extinguisher.
-
Battery Fire: Damaged lithium battery may ignite; use Class D extinguisher or sand.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties