Infant T-Piece Resuscitator
WhatsApp Order
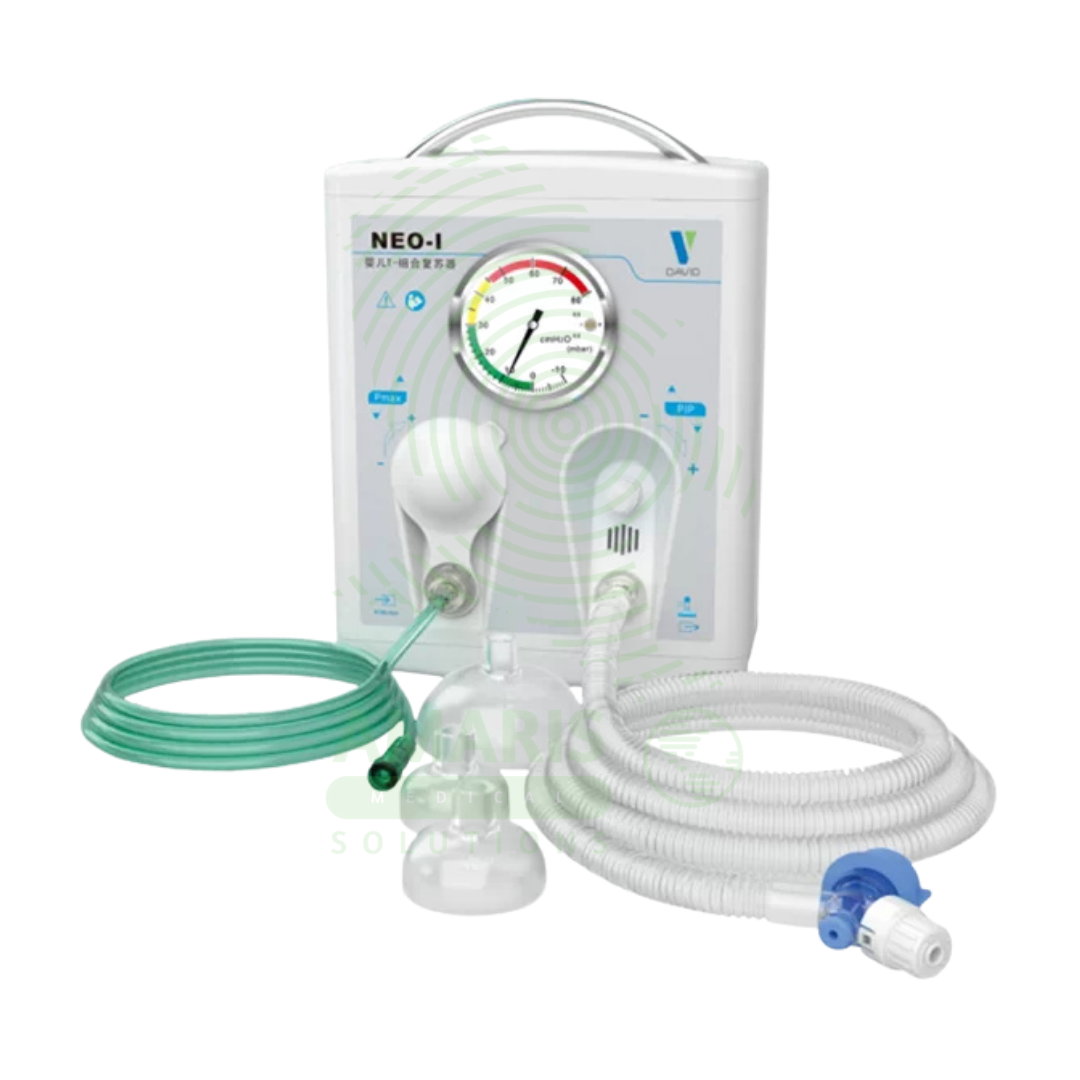
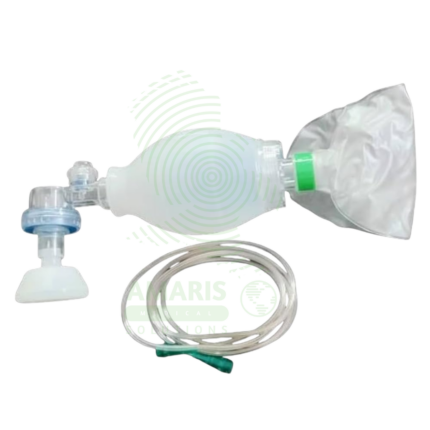
An Infant T-piece Resuscitator is a Class II medical device designed for neonatal resuscitation and controlled positive pressure ventilation, delivering precise, consistent peak inspiratory pressures (PIP) and positive end-expiratory pressure (PEEP) as recommended by Neonatal Resuscitation Program (NRP) guidelines. The T-piece assembly connects to compressed air/oxygen source (5-15 L/min) with adjustable PIP (10-40 cmH₂O) and PEEP (0-10 cmH₂O) controls, built-in pressure manometer, and pressure relief valve (30-40 cmH₂O). Operator occludes port to deliver breaths (typically 40-60/min), with consistent pressures maintained regardless of technique. Essential advantages over bag-valve-mask devices include operator-independent pressure delivery, ability to provide PEEP (critical for preterm lung protection), reduced risk of barotrauma, and hands-free PEEP allowing two-handed mask seal. Primary clinical applications include delivery room resuscitation, stabilization of extremely preterm infants (<1,000 g), CPAP/PEEP delivery, meconium aspiration management, and neonatal transport ventilation. Critical safety considerations include verifying gas supply (5-15 L/min), monitoring manometer pressures, ensuring proper mask seal, observing chest rise, and titrating FiO2 to SpO2 targets. Indispensable resuscitation equipment in every delivery room, NICU, and neonatal transport setting worldwide.
Categories: ANESTHESIA AND RESPIRATORY EQUIPMENT, Airway Management, EMERGENCY AND FIRST AID KIT, Emergency Resuscitation Equipment, OBSTETRICS, GYNECOLOGY AND NEONATAL
Tags: Delivery Room Equipment, Infant-T piece Resuscitator, Neonatal CPAP, Neonatal Resuscitator, Neonatal Ventilation, NRPResuscitator, PEEP Resuscitator, T-piece Resuscitator
Description
Infant T-Piece Resuscitator
PRIMARY CLINICAL & DIAGNOSTIC USES
1. Neonatal Resuscitation in Delivery Rooms
-
Primary Use: The standard device for providing positive-pressure ventilation to newborns requiring respiratory support at birth, as recommended by the Neonatal Resuscitation Program (NRP) and ILCOR guidelines.
-
How it helps: For the neonatologist and delivery room team, it provides a guideline-endorsed, reliable tool that standardizes resuscitation across different providers and institutions. For the newborn taking their first breaths, it means they receive the highest standard of evidence-based care from the very first moment they need support, giving them the best possible start.
2. Controlled Positive Pressure Ventilation
-
Primary Use: Delivers precise, consistent peak inspiratory pressure (PIP) and positive end-expiratory pressure (PEEP) during manual ventilation, reducing variability compared to bag-valve-mask devices.
-
How it helps: For the respiratory therapist and resuscitation team, it eliminates the guesswork of manual ventilation—set the pressures once, and every breath is identical regardless of who is squeezing the bag. For the fragile infant, particularly preterm babies, this consistency is life-saving: it prevents the wildly varying pressures that can rupture fragile lungs (barotrauma) while ensuring each breath effectively opens the airways.
3. CPAP and PEEP Delivery
-
Primary Use: Provides continuous positive airway pressure (CPAP) or PEEP to stabilize alveoli, maintain functional residual capacity, and improve oxygenation in spontaneously breathing but distressed newborns.
-
How it helps: For the clinician, the ability to deliver PEEP with a manual device keeps small air sacs (alveoli) from collapsing between breaths, a feature traditional bags often lack. For the infant working hard to breathe, this constant pressure means each spontaneous breath requires less effort, preserving their limited energy for growth and recovery rather than exhausting themselves just to keep their lungs open.
4. Resuscitation of Extremely Preterm Infants
-
Primary Use: Essential for ventilating extremely low birth weight infants (<1,000 g) where precise pressure control is critical to prevent barotrauma, pneumothorax, and bronchopulmonary dysplasia.
-
How it helps: For the neonatal team caring for a 500-gram micro-preemie, the T-piece’s precision is non-negotiable—pressures that would be safe for a full-term baby can instantly destroy the lungs of an extremely preterm infant. For this tiniest of patients, the device’s ability to deliver gentle, controlled breaths means they have a fighting chance to survive without the devastating lung complications that plagued previous generations of preemies.
5. Meconium-Stained Amniotic Fluid Management
-
Primary Use: Used to provide positive-pressure ventilation after intubation and tracheal suctioning in infants born through meconium-stained fluid who are not breathing effectively.
-
How it helps: For the resuscitation team managing this high-risk scenario, the T-piece delivers consistent ventilation while minimizing the risk of pushing meconium deeper into the airways. For the infant who has aspirated meconium, controlled ventilation with PEEP helps keep compromised airways open while protecting delicate lung tissue from further injury.
6. Transport Ventilation
-
Primary Use: Portable T-piece resuscitators are used during neonatal transport to provide consistent ventilation with controlled pressures, PEEP, and FiO₂ while moving infants between facilities.
-
How it helps: For the transport team moving a fragile infant in an ambulance or helicopter, the T-piece maintains ICU-level precision regardless of vehicle vibration or movement. For the critically ill newborn being transferred to a specialized center, it ensures the same quality of ventilation during transport that they received in the referring hospital, protecting them during the most vulnerable part of their journey.
7. Stabilization Before Surfactant Administration
-
Primary Use: Delivers controlled ventilation before, during, and after surfactant instillation for preterm infants with respiratory distress syndrome.
-
How it helps: For the neonatologist administering surfactant, the T-piece maintains lung recruitment during the procedure, ensuring the medication distributes evenly throughout the lungs. For the preterm infant receiving this life-saving therapy, consistent ventilation during surfactant administration means maximum benefit from the medication and minimal risk of complications during this critical intervention.
SECONDARY & SUPPORTIVE USES
1. Simulation and Training: For the NRP instructor, the T-piece is the standard tool for teaching neonatal ventilation. For the trainee—whether a new nurse, resident, or respiratory therapist—mastering this device builds competence and confidence that will directly translate to better outcomes for their tiniest future patients.
2. Research and Clinical Studies: For the neonatal researcher, it provides a standardized method of delivering ventilation in clinical studies. For future generations of infants, this consistency in research protocols leads to clearer data and improved resuscitation guidelines.
3. Quality Improvement Programs: For the quality and safety team, the T-piece reduces variability between providers, making ventilation more consistent across an entire institution. For every infant born in that hospital, this standardization means they receive the same high-quality resuscitation regardless of which team member is at the bedside.
4. Neonatal Intensive Care Unit (NICU) Ventilation: For the NICU team, it provides reliable short-term ventilation when mechanical ventilators are being set up or during bedside procedures. For the infant in the NICU, it ensures uninterrupted respiratory support during transitions, protecting them from destabilizing events.
5. Community Hospital and Resource-Limited Settings: For the community hospital without sophisticated mechanical ventilators, the T-piece provides reliable, pressure-limited ventilation for newborns in distress. For families in rural or underserved areas, it means their baby can receive high-quality resuscitation close to home, without the need for immediate transfer to a distant center.
6. Delivery Room Preparedness: For the hospital administration and obstetrics team, having a T-piece at every resuscitation station ensures readiness for any delivery complication. For every mother entering that delivery room, it provides reassurance that the team is prepared to support her baby if the unexpected happens.
KEY PRODUCT FEATURES
1. BASIC IDENTIFICATION ATTRIBUTES
-
Product Type: Manual resuscitator device specifically designed for neonatal use, delivering controlled positive pressure ventilation with adjustable PIP, PEEP, and FiO2.
-
Common Names: Infant T-piece Resuscitator, T-piece Resuscitator, Neonatal T-piece, T-piece Ventilator, Pressure-Limited Resuscitator, NRP Resuscitator.
-
Components:
-
T-Piece Assembly: T-shaped connector with gas inlet, patient outlet, and pressure control ports.
-
Pressure Relief Valve: Adjustable or fixed valve to limit maximum inspiratory pressure (typically 30-40 cmH₂O).
-
PEEP Valve: Adjustable valve to maintain positive end-expiratory pressure (typically 0-10 cmH₂O).
-
Gas Supply Tubing: Connects to compressed air and oxygen source or blender.
-
Patient Connection: Standard 15 mm/22 mm connector for endotracheal tube, mask, or nasal prongs.
-
Pressure Manometer: Built-in or detachable gauge displaying airway pressure in cmH₂O.
-
Oxygen Reservoir/Blender Connection: Port for connecting to air/oxygen blender for precise FiO2 control.
-
-
Controls:
-
Inspiratory Pressure Control: Adjusts PIP (typically 10-40 cmH₂O).
-
PEEP Control: Adjusts end-expiratory pressure (typically 0-10 cmH₂O).
-
Occlusion Port: Finger-operated port to occlude for delivering breaths.
-
-
Pressure Range: 0-40 cmH₂O (inspiratory); 0-10 cmH₂O (PEEP).
-
Gas Supply: Requires compressed medical air and oxygen (40-60 psi) or blended gas source.
-
Flow Rate: Typically 5-15 L/min (adjustable based on patient size and needs).
2. TECHNICAL & PERFORMANCE PROPERTIES
-
Pressure Accuracy: ±2-5 cmH₂O of set pressure (varies by device).
-
PEEP Maintenance: Consistent PEEP regardless of breath rate or operator technique.
-
Pressure Relief: Safety valve prevents excessive pressure (typically set at 30-40 cmH₂O).
-
Gas Consumption: 5-15 L/min continuous flow (higher flow rates may be needed for larger infants).
-
FiO2 Delivery: 21-100% when connected to air/oxygen blender.
-
Temperature Stability: Gas may be heated and humidified for prolonged use (in NICU settings).
-
Manometer Response: Real-time pressure display with minimal lag.
-
Dead Space: Minimal when properly configured.
-
Operating Temperature: 10-40°C (50-104°F).
-
Humidity: Can be used with heated humidifiers for extended ventilation.
3. PHYSICAL & OPERATIONAL PROPERTIES
-
Dimensions (T-piece assembly): 10-15 cm × 5-8 cm × 3-5 cm.
-
Weight: 200-500 grams (complete assembly with tubing).
-
Materials: Medical-grade plastics (polycarbonate, ABS), silicone seals, stainless steel components.
-
Pressure Manometer: Analog gauge (typically 0-60 cmH₂O) or digital display on some models.
-
Tubing: Flexible, kink-resistant PVC or silicone (length 1.5-2.5 m).
-
Connectors: Standard 15 mm/22 mm ISO tapered fittings.
-
Color Coding: Often color-coded for neonatal use (typically blue or clear).
-
Packaging: Sterile (for single-use) or non-sterile (reusable) available.
-
Accessories: Face masks (various sizes), endotracheal tube connectors, nasal prongs.
4. SAFETY & COMPLIANCE ATTRIBUTES
-
Regulatory Status: Class II medical device requiring FDA 510(k) clearance; CE marked.
-
Clinical Standards: Complies with ISO 10651-5 (resuscitators) and ISO 80601-2-12 (critical care ventilators - relevant standards).
-
Pressure Limitation: Built-in pressure relief valve prevents excessive inspiratory pressures.
-
PEEP Stability: Maintains set PEEP regardless of operator technique, preventing alveolar collapse.
-
Gas Supply Failure: Some models include alarms for gas supply interruption.
-
Material Safety: Medical-grade, biocompatible, latex-free materials.
-
Electrical Safety: Not applicable (pneumatic device, no electrical components).
-
Biocompatibility: All patient-contact materials meet ISO 10993 standards.
-
Cleaning/Sterilization: Reusable models withstand sterilization (autoclave, ETO) per manufacturer.
-
Single-Use Options: Disposable, sterile units available for infection control.
-
Quality Management: Manufactured under ISO 13485 certified processes.
5. STORAGE & HANDLING ATTRIBUTES
-
Storage: Store in a clean, dry environment; protect from dust, extreme temperatures, and physical damage.
-
Assembly: Connect to gas source (air/oxygen blender or flow meter); verify all connections secure.
-
Pressure Setting: Adjust PIP and PEEP according to clinical protocol (NRP guidelines: initial PIP 20-25 cmH₂O, PEEP 5 cmH₂O).
-
Leak Testing: Before use, test system for leaks by occluding patient port and observing pressure.
-
Cleaning (Reusable): Disassemble; clean with mild detergent; rinse thoroughly; sterilize per manufacturer instructions.
-
Disposable Units: Single-use only; discard after use; do not resterilize or reuse.
-
Inspection: Before each use, check for cracks, damaged tubing, proper valve function, and accurate pressure readings.
-
Expiration: Check expiration date on disposable units; do not use expired devices.
-
Emergency Preparedness: Keep assembled and ready in delivery rooms, NICUs, and transport equipment.
6. LABORATORY & CLINICAL APPLICATIONS
-
Primary Application: Neonatal resuscitation and controlled positive pressure ventilation.
-
Neonatal Resuscitation Program (NRP) Guidelines:
-
Initial Ventilation: PIP 20-25 cmH₂O, PEEP 5 cmH₂O, rate 40-60 breaths/min.
-
Adjustment: Increase PIP if chest not rising; decrease if overinflation.
-
PEEP: Maintains lung recruitment; essential for preterm infants.
-
Oxygen: Start with 21% (term) or 21-30% (preterm); titrate to SpO2 targets.
-
-
Ventilation Technique:
-
Set PIP and PEEP according to clinical situations.
-
Connect to a gas source (blender or flow meter) at 5-15 L/min.
-
Apply a mask to the infant's face or connect to ETT.
-
Occlusion of T-piece port delivers inspiratory breath at set PIP.
-
Release occlusion allows exhalation; PEEP maintained.
-
Rate controlled by operator (typically 40-60 breaths/min).
-
Advantages Over Bag-Valve-Mask:
-
Consistent PIP and PEEP (operator-independent).
-
Reduced variability in delivered pressures.
-
Ability to deliver PEEP (critical for preterm lung protection).
-
Lower risk of barotrauma and volutrauma.
-
Hands-free PEEP allows the operator to maintain a mask seal with both hands.
-
-
Transition to Mechanical Ventilation: Can be used to provide manual ventilation while setting up mechanical ventilators.
SAFETY HANDLING PRECAUTIONS
1. SAFETY PRECAUTIONS
-
Pressure Monitoring: Always observe the manometer during ventilation; verify set pressures are delivered.
-
Gas Supply: Ensure adequate gas flow (5-15 L/min) to maintain set pressures; low flow may cause inadequate ventilation.
-
PEEP Setting: Use PEEP (5 cmH₂O) for all preterm infants and term infants requiring ventilation; prevents alveolar collapse.
-
Pressure Relief: Verify pressure relief valve is set appropriately (typically 30-40 cmH₂O) to prevent barotrauma.
-
Mask Seal: Maintain good seal to deliver set pressures; leak around mask reduces effective ventilation.
-
Chest Rise: Observe chest rise to assess adequacy of ventilation; adjust PIP as needed.
-
Oxygen Titration: Use blended oxygen; start with low FiO2 and titrate to SpO2 targets (prevents hyperoxia).
-
Endotracheal Tube Position: If using ETT, verify proper position (chest rise, auscultation, CO2 detector).
-
Equipment Check: Verify function before each use; have backup device available.
-
Training: All personnel should be trained in T-piece resuscitator use per NRP guidelines.
2. FIRST AID MEASURES
-
Inadequate Ventilation: Check gas supply, mask seal, airway position; increase PIP if needed; consider intubation.
-
Equipment Failure: Switch to backup resuscitation device (bag-valve-mask) immediately; replace faulty device.
-
Gas Supply Failure: Disconnect; use resuscitation bag with room air or backup oxygen source.
-
Patient Deterioration: Assess for pneumothorax, equipment malfunction, or need for advanced interventions.
3. FIRE FIGHTING MEASURES
-
Flammability: Plastic components are combustible; oxygen supports combustion.
-
Extinguishing Media: Use CO₂ or dry chemical extinguisher for electrical fires; water for surrounding materials.
-
Oxygen Source: Shut off oxygen if fire is suspected.


 Dermatoscope and Magnifiers
Dermatoscope and Magnifiers Diagnostic Kits
Diagnostic Kits Vital Signs Monitors
Vital Signs Monitors Stethoscopes and Accessories
Stethoscopes and Accessories Otoscopes, Ophthalmoscopes, and Retinoscopes
Otoscopes, Ophthalmoscopes, and Retinoscopes Reflex Hammers and Neurological Tools
Reflex Hammers and Neurological Tools Scales and Measuring Devices
Scales and Measuring Devices Spirometers and Pulmonary Function Tests
Spirometers and Pulmonary Function Tests
 Electrosurgical Units and Accessories
Electrosurgical Units and Accessories Cutting Instruments
Cutting Instruments Grasping and Holding Instruments
Grasping and Holding Instruments Hemostatic Instruments
Hemostatic Instruments Specialized Surgical Sets
Specialized Surgical Sets Single-Use Procedure Trays and Packs
Single-Use Procedure Trays and Packs Surgical Drapes, Gowns, and Covers
Surgical Drapes, Gowns, and Covers Tissue Unifying Instruments
Tissue Unifying Instruments
 Radiation Protection
Radiation Protection X-Ray Machines and Accessories
X-Ray Machines and Accessories Ultrasound Systems and Probes
Ultrasound Systems and Probes MRI and CT Scanners
MRI and CT Scanners Radiology Consumables
Radiology Consumables Bone Densitometers
Bone Densitometers Fluoroscopy Equipment
Fluoroscopy Equipment Imaging Tables and Positioning Aids
Imaging Tables and Positioning Aids
 Microscopes and Accessories
Microscopes and Accessories Centrifuges and Separators
Centrifuges and Separators Analyzers
Analyzers Incubators and Ovens
Incubators and Ovens Pipettes, Dispensers, and Lab Glassware
Pipettes, Dispensers, and Lab Glassware Refrigerators, Freezers, and Storage Units
Refrigerators, Freezers, and Storage Units Lab Consumables
Lab Consumables Sterilizers and Autoclaves for Lab Use
Sterilizers and Autoclaves for Lab Use
 Multi-Parameter Monitors
Multi-Parameter Monitors Ventilators and Respiratory Support Devices
Ventilators and Respiratory Support Devices Defibrillators and AEDs
Defibrillators and AEDs Infusion Pumps and IV Systems
Infusion Pumps and IV Systems Patient Warmers and Cooling Devices
Patient Warmers and Cooling Devices Central Monitoring Stations
Central Monitoring Stations Accessories
Accessories
 Anesthesia Machines and Workstations
Anesthesia Machines and Workstations Oxygen Concentrators and Delivery Systems
Oxygen Concentrators and Delivery Systems Nebulizers and Inhalers
Nebulizers and Inhalers CPAP/BiPAP Machines
CPAP/BiPAP Machines Airway Management
Airway Management Anesthesia Masks, Circuits, and Bags
Anesthesia Masks, Circuits, and Bags Humidifiers and Heaters
Humidifiers and Heaters Respiratory Therapy Accessories
Respiratory Therapy Accessories
 First Aid Kits and Cabinets
First Aid Kits and Cabinets Emergency Resuscitation Equipment
Emergency Resuscitation Equipment Trauma Supplies
Trauma Supplies Emergency Carts and Crash Carts
Emergency Carts and Crash Carts Burn Care Products
Burn Care Products Bleeding Control
Bleeding Control Automated External Defibrillators (AEDs)
Automated External Defibrillators (AEDs) Transport and Evacuation
Transport and Evacuation
 Wheelchairs and Accessories
Wheelchairs and Accessories Walkers, Crutches, and Canes
Walkers, Crutches, and Canes Prosthetics and Orthotics
Prosthetics and Orthotics Physical Therapy Equipment
Physical Therapy Equipment Transfer Devices
Transfer Devices Bathroom Safety
Bathroom Safety Orthopedic Traction and Tables
Orthopedic Traction and Tables Hot/Cold Therapy Packs and Units
Hot/Cold Therapy Packs and Units
 Beds and Mattresses
Beds and Mattresses Chairs and Stools
Chairs and Stools Tables
Tables Cabinets and Storage
Cabinets and Storage Privacy Screens & Curtains
Privacy Screens & Curtains Stands and Racks
Stands and Racks Linens and Textiles
Linens and Textiles Lighting
Lighting
 Autoclaves and Sterilizers
Autoclaves and Sterilizers Ultrasonic Cleaners
Ultrasonic Cleaners Disinfectant Solutions and Wipes
Disinfectant Solutions and Wipes Sterilization Pouches, Wraps, and Indicators
Sterilization Pouches, Wraps, and Indicators Instrument Trays and Containers
Instrument Trays and Containers UV and Ozone Disinfection Devices
UV and Ozone Disinfection Devices Washer Disinfectors
Washer Disinfectors
 Wound Care
Wound Care Gloves
Gloves Masks and Respirators
Masks and Respirators Catheters and Tubing
Catheters and Tubing Swabs, Applicators, and Sponges
Swabs, Applicators, and Sponges Incontinence Products
Incontinence Products Personal Protective Equipment (PPE)
Personal Protective Equipment (PPE)
 Dental Chairs and Units
Dental Chairs and Units Handpieces and Burs
Handpieces and Burs Instruments
Instruments Consumables
Consumables Sterilization for Dental Use
Sterilization for Dental Use Orthodontic Supplies
Orthodontic Supplies Endodontic Tools
Endodontic Tools
 Slit Lamps and Tonometers
Slit Lamps and Tonometers Lensometers and Phoropters
Lensometers and Phoropters Ophthalmic Surgical Instruments
Ophthalmic Surgical Instruments Eyewear Frames and Lenses
Eyewear Frames and Lenses Contact Lens Supplies
Contact Lens Supplies Vision Testing Charts and Devices
Vision Testing Charts and Devices Eye Care Consumables
Eye Care Consumables Laser Systems for Eye Care
Laser Systems for Eye Care
 ENT Exam Chairs and Tables
ENT Exam Chairs and Tables Endoscopes
Endoscopes Audiometers and Hearing Tests
Audiometers and Hearing Tests ENT Instruments
ENT Instruments Nasal and Throat Packs
Nasal and Throat Packs Hearing Aids and Accessories
Hearing Aids and Accessories Otology Supplies
Otology Supplies
 Fetal Dopplers and Monitors
Fetal Dopplers and Monitors Delivery Beds and Tables
Delivery Beds and Tables Gynecological Instruments
Gynecological Instruments Neonatal Incubators and Warmers
Neonatal Incubators and Warmers Breast Pumps and Accessories
Breast Pumps and Accessories Contraceptive Devices
Contraceptive Devices Maternity Supports and Pads
Maternity Supports and Pads Neonatal Consumables
Neonatal Consumables
 Cystoscopes and Urethroscopes
Cystoscopes and Urethroscopes Dialysis Machines and Supplies
Dialysis Machines and Supplies Urological Catheters and Bags
Urological Catheters and Bags Lithotripters
Lithotripters Prostate Treatment Devices
Prostate Treatment Devices Urinary Incontinence Products
Urinary Incontinence Products Kidney Stone Management Tools
Kidney Stone Management Tools Consumables & Disposables
Consumables & Disposables
 EEG and EMG Machines
EEG and EMG Machines Neurosurgical Instruments
Neurosurgical Instruments Nerve Stimulators
Nerve Stimulators Headrests and Positioning Aids
Headrests and Positioning Aids Lumbar Puncture Kits
Lumbar Puncture Kits Seizure Monitoring Devices
Seizure Monitoring Devices Consumables
Consumables Rehabilitation for Neurological Conditions
Rehabilitation for Neurological Conditions
 ECG Machines and Accessories
ECG Machines and Accessories Holter Monitors
Holter Monitors Stress Test Systems
Stress Test Systems Pacemakers and Defibrillator Accessories
Pacemakers and Defibrillator Accessories Vascular Access Devices
Vascular Access Devices Cardiac Catheters and Guidewires
Cardiac Catheters and Guidewires Blood Flow Meters
Blood Flow Meters Consumables
Consumables
 Orthopedic Instruments
Orthopedic Instruments Casts, Splints, and Padding
Casts, Splints, and Padding Joint Replacement Supplies
Joint Replacement Supplies Prosthetic Limbs and Components
Prosthetic Limbs and Components Bone Grafts and Substitutes
Bone Grafts and Substitutes Traction Devices
Traction Devices Orthopedic Braces and Supports
Orthopedic Braces and Supports Rehabilitation Aids for Orthopedics
Rehabilitation Aids for Orthopedics
 Home Oxygen Therapy
Home Oxygen Therapy Hospital Beds for Home Use
Hospital Beds for Home Use Mobility Aids
Mobility Aids Bathroom and Daily Living Aids
Bathroom and Daily Living Aids Wound Care for Home
Wound Care for Home Monitoring Devices
Monitoring Devices Enteral Feeding Pumps and Tubes
Enteral Feeding Pumps and Tubes
 Hand Sanitizers and Dispensers
Hand Sanitizers and Dispensers Face Shields and Goggles
Face Shields and Goggles Isolation Gowns and Suits
Isolation Gowns and Suits Biohazard Waste Containers
Biohazard Waste Containers Air Purifiers and HEPA Filters
Air Purifiers and HEPA Filters Surface Disinfectants
Surface Disinfectants Sharps Containers
Sharps Containers Protective Barriers
Protective Barriers
 Cardiovascular & Endurance Training
Cardiovascular & Endurance Training Strength Training & Weightlifting
Strength Training & Weightlifting Functional Training & Core Conditioning
Functional Training & Core Conditioning Physical Therapy & Rehabilitation
Physical Therapy & Rehabilitation Sports & Outdoor Recreation
Sports & Outdoor Recreation Gym Flooring & Facility Equipment
Gym Flooring & Facility Equipment Fitness Monitoring & Accessories
Fitness Monitoring & Accessories Kids & Novelties
Kids & Novelties